I was once the HSE Manager for a remote mining project housing 2,000 workers where the nearest hospital was a four-hour flight away. One morning, three workers reported to the clinic with severe gastrointestinal distress; by noon, that number hit thirty. We triggered our emergency response plan, not for an explosion or a collapse, but for a breakdown in our catering supply chain. The investigation didn’t reveal a “bad batch” of chicken—it revealed a systemic failure in monitoring refrigeration temperatures during transport. That incident nearly shut down a billion-dollar operation because we failed to manage biological hazards at a critical control point.
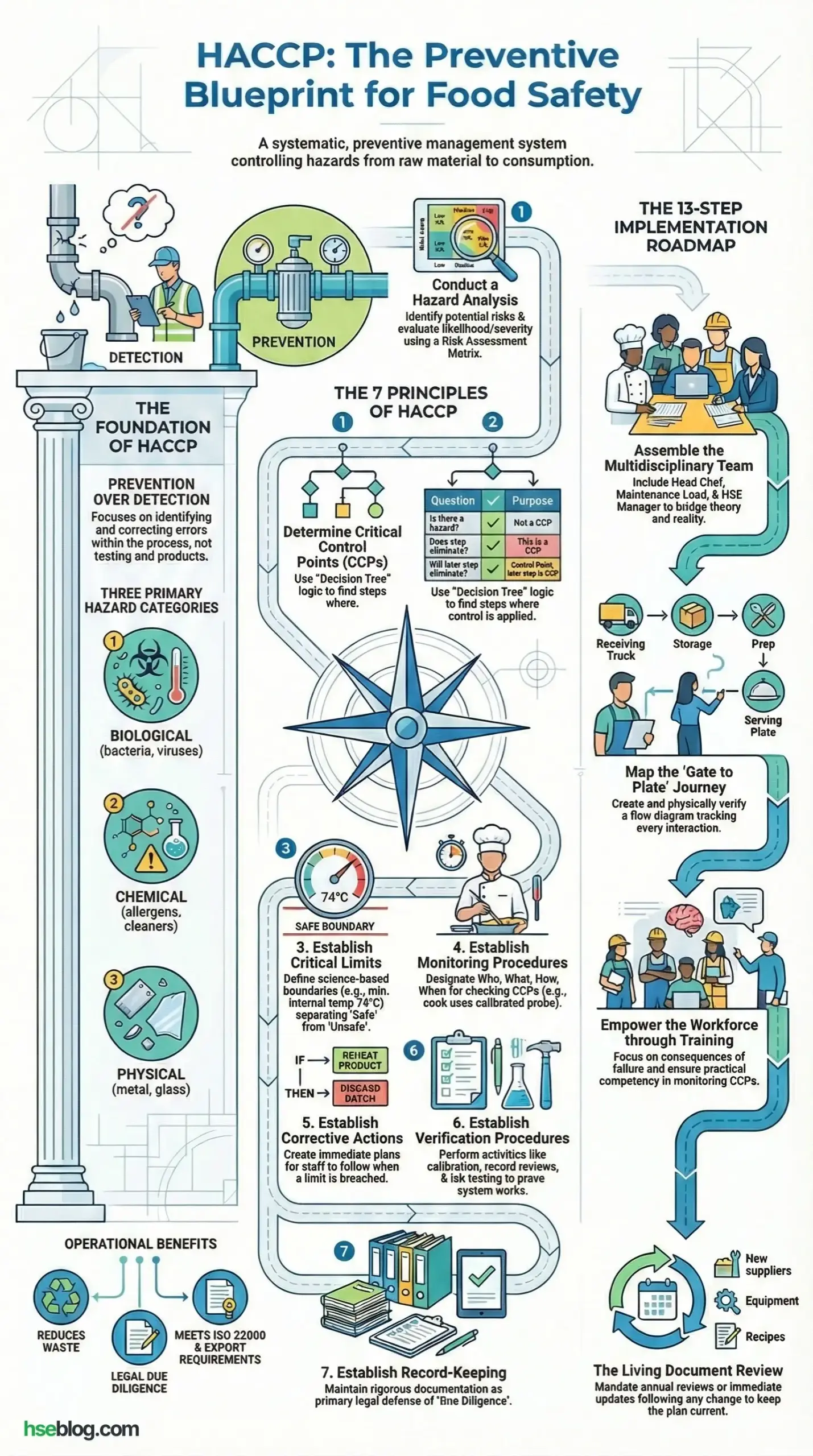
This is why HACCP (Hazard Analysis and Critical Control Points) is critical. It is not just a food safety certificate for the wall; it is a systematic, preventive approach to physical, chemical, and biological safety. In high-risk industries, whether we are managing an offshore rig canteen or a chemical processing plant, HACCP provides the logic to identify where things can go wrong and put hard stops in place to prevent them. This article defines the HACCP methodology, details its seven principles, and explains how to implement it effectively to protect human life.
TL;DR
- Prevention over detection: HACCP stops hazards before they reach the plate; you cannot “inspect quality” into a product at the end of the line.
- Critical Control Points (CCPs) are non-negotiable: These are the specific stages (like cooking temperature or cooling time) where a loss of control results in an unacceptable health risk.
- Documentation defends the operation: In an outbreak investigation or audit, if a temperature check isn’t recorded, legally and practically, it never happened.
- It applies beyond food factories: I have applied HACCP principles to potable water systems, camp catering in mining sites, and waste management to prevent biological hazards.
What is HACCP? Definition and Meanings
HACCP stands for Hazard Analysis and Critical Control Points. It is an internationally recognized management system in which food safety is addressed through the analysis and control of biological, chemical, and physical hazards from raw material production, procurement, and handling, to manufacturing, distribution, and consumption.
From a field perspective, HACCP is risk assessment applied specifically to the flow of consumable products. Unlike traditional quality control, which tests the end product (reactive), HACCP is designed to detect and correct errors in the process (preventive).
When I audit a facility, I look for HACCP as the backbone of their safety culture. It means the organization acknowledges that hazards exist and has engineered a process to eliminate or reduce them to an acceptable level before they ever impact the consumer.
Importance Of HACCP
The importance of HACCP goes far beyond regulatory compliance. In my experience managing remote sites, a foodborne illness outbreak is an operational disaster. It affects morale, halts productivity, and opens the company up to massive liability.
Key operational benefits include:
- Risk Reduction: It systematically identifies hazards (like Salmonella in poultry or metal shards in ground meat) and creates a barrier against them.
- Due Diligence: In the event of legal action, a robust HACCP plan with verified records is your primary defense that you took all reasonable precautions.
- Cost Savings: It reduces product waste. If you catch a temperature deviation early (monitoring), you can fix it before you have to discard a whole production run.
- Global Standard: It is compatible with ISO 22000 and is a requirement for exporting food products to most international markets, including the US (FDA) and EU.
7 Key Principles Of HACCP (Hazard Analysis and Critical Control Points)
As an auditor, this is the section I scrutinize the most. If these principles are not applied correctly, your safety plan is just a paper tiger—it looks good on a shelf but won’t stop a real hazard on the production line. I have written this based on the Codex Alimentarius standards but added the practical “field reality” I expect to see when I inspect a high-risk facility.
Principle 1: Conduct a Hazard Analysis

This is the foundation of the entire system. You cannot control a risk you haven’t identified. In this phase, we act as investigators, looking at the process flow to find where things could go wrong. We don’t just guess; we use data, experience, and on-site observation.
We look for three specific types of hazards:
- Biological Hazards: These are the most dangerous in camp catering and food processing. They include bacteria (Salmonella, Listeria, E. coli), viruses (Norovirus, Rotavirus), and parasites.
- Field Example: Thawing meat on a counter at room temperature allows dormant bacteria to multiply rapidly.
- Chemical Hazards: These can be naturally occurring (allergens like peanuts or shellfish), intentionally added (excessive preservatives), or accidental contaminants.
- Field Example: A cleaner using an industrial degreaser on a food prep surface without rinsing it properly.
- Physical Hazards: Foreign objects that can cause injury, choking, or dental damage.
- Field Example: Metal shavings from a dull can opener, glass from a shattered lightbulb, or plastic bristles from a cleaning brush.
The Risk Assessment Matrix:
For every hazard found, we must evaluate:
- Likelihood: How often will this happen?
- Severity: If it happens, will it kill someone or just give them a stomach ache?Only significant hazards require a Critical Control Point.
Principle 2: Determine Critical Control Points (CCPs)
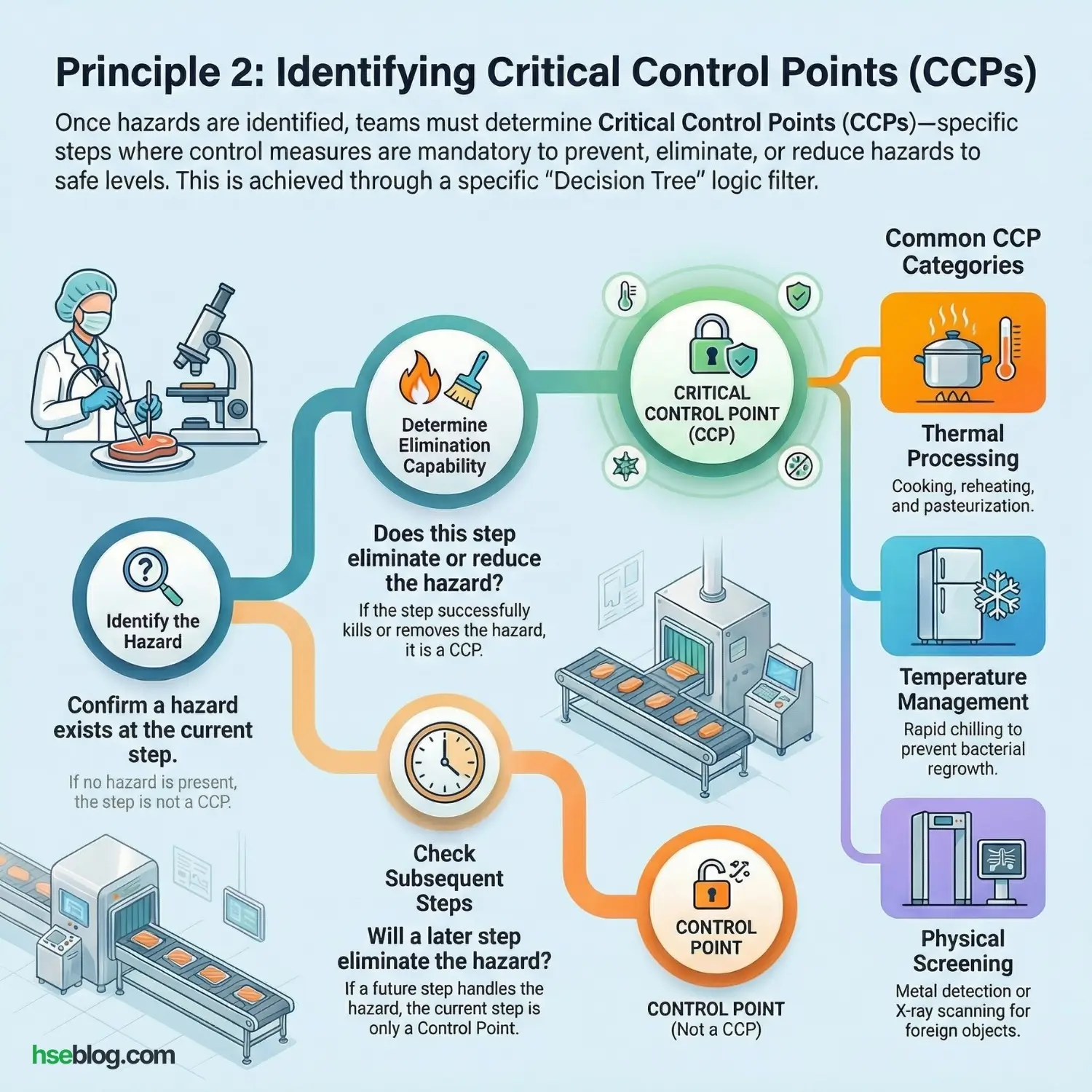
Once hazards are identified, we must decide where to stop them. A Critical Control Point (CCP) is a step in the process where a control measure must be applied to prevent, eliminate, or reduce a hazard to an acceptable level.
The “Decision Tree” Logic:
To decide if a step is a CCP, I ask my teams to use a logic filter:
- Is there a hazard at this step? (If no, not a CCP).
- Does this step eliminate the hazard? (e.g., Pasteurization kills bacteria $\rightarrow$ Yes, this is a CCP).
- Will a later step eliminate the hazard? (e.g., Storing raw beef is risky, but we will cook it later. So, storage is a Control Point, but Cooking is the Critical Control Point).
Common CCPs include:
- Thermal Processing: Cooking, reheating, pasteurization.
- Chilling: Rapid cooling to prevent bacterial regrowth.
- Testing: Metal detection or X-ray scanning.
Principle 3: Establish Critical Limits
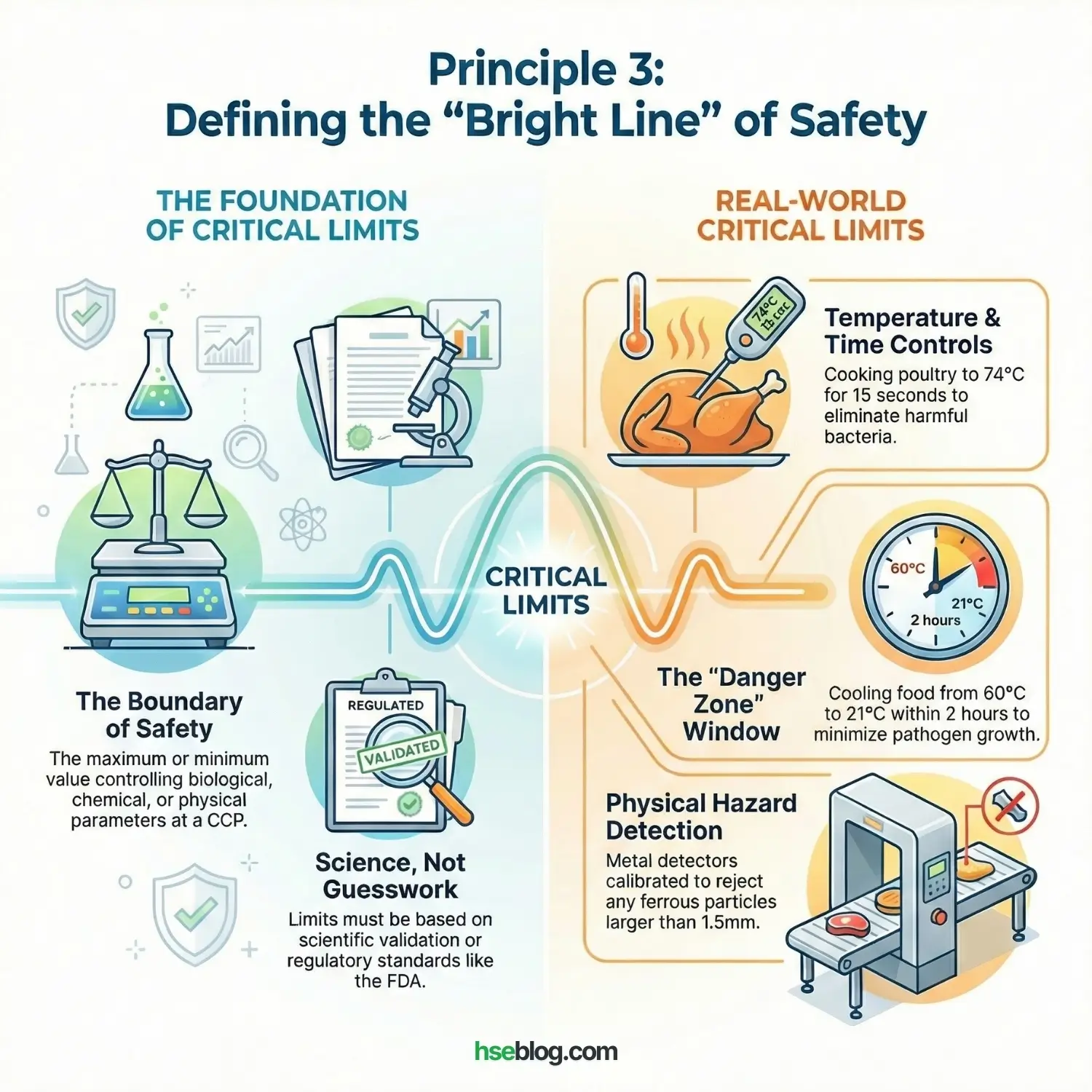
A CCP is useless without a limit. A Critical Limit is the maximum or minimum value to which a biological, chemical, or physical parameter must be controlled at a CCP. It draws a bright line between “Safe” and “Unsafe.”
Science, Not Guesswork:
These limits must be based on regulatory standards (like FDA or local Food Authority) or scientific validation.
- Temperature: “Minimum internal temperature of 74°C for 15 seconds” (to kill poultry bacteria).
- Time: “Cool from 60°C to 21°C within 2 hours” (to minimize time in the ‘Danger Zone’).
- Physical: “Metal detector must reject any ferrous particle larger than 1.5mm.”
I often advise sites to set an “Operating Limit” that is stricter than the Critical Limit. If the Critical Limit is 74°C, set your machine to 76°C. This gives you a safety buffer so that normal process variations don’t trigger a safety violation.
Principle 4: Establish Monitoring Procedures

Monitoring is the act of conducting a planned sequence of observations or measurements to assess whether a CCP is under control. It answers the question: “Is the process working right now?”
The 4 Elements of Monitoring:
- Who: The person responsible (e.g., The Line Cook, NOT “The Kitchen Staff”).
- What: The attribute being measured (e.g., Internal temperature of the chicken breast).
- How: The method used (e.g., Using a calibrated probe thermometer inserted into the thickest part of the meat).
- When: The frequency (e.g., Every batch, or every 30 minutes).
Types of Monitoring:
- Continuous: Automated temperature recorders in freezers that alarm if the temp rises.
- Discontinuous: A manual check by a worker. This relies heavily on training and integrity.
Principle 5: Establish Corrective Actions

What happens when the monitoring shows the Critical Limit has been breached? You cannot wait for a management meeting. The floor staff must take immediate Corrective Action.
Two Parts to Corrective Action:
- Fix the Process: Bring the CCP back under control immediately.
- Example: “The oven is only at 150°C. Turn it up to 180°C and extend the cooking time.”
- Handle the Product: Decide what to do with the food produced while the process was out of control.
- Example: “The milk pasteurizer dropped below 72°C. Divert all milk processed in the last 15 minutes to the drain. Do not bottle it.”
Documentation: Every corrective action must be recorded. I want to see records of problems. If a site has zero corrective actions recorded in a year, I suspect they are faking their logs.
Principle 6: Establish Verification Procedures
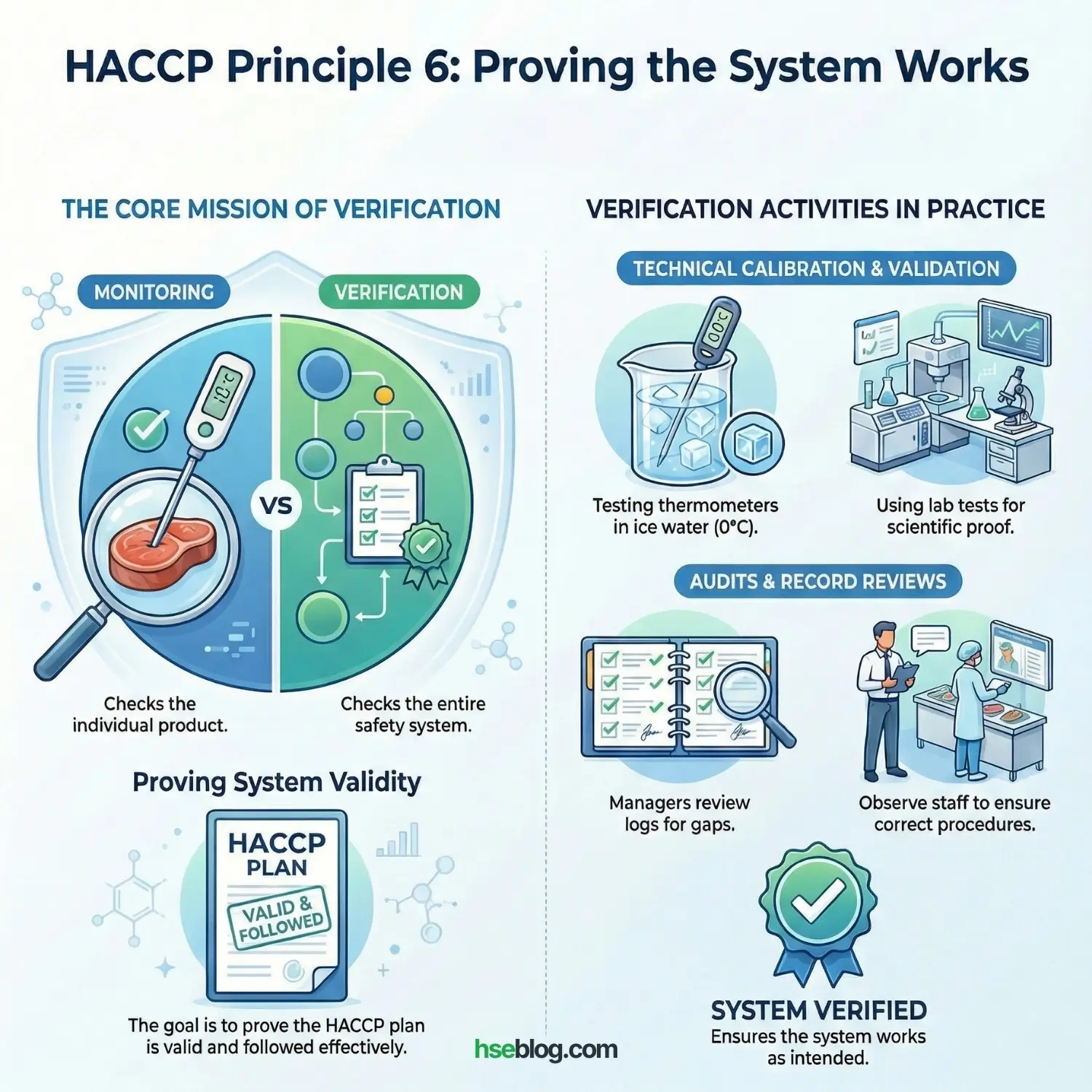
Monitoring checks the product; Verification checks the system. We need to prove that the HACCP plan is actually valid and is being followed effectively.
Verification Activities Include:
- Calibration: Checking thermometers against a standard (ice water slurry at 0°C) to ensure they are accurate.
- Record Review: An HSE Manager reviewing the weekly logs to ensure no gaps exist and signatures are real.
- Validation: Scientific proof. Sending a sample of your cooked product to a lab to confirm that your cooking time/temp actually killed the bacteria (Salmonella/Listeria) as intended.
- Internal Audits: Watching the staff to ensure they are actually performing the monitoring steps correctly.
Principle 7: Establish Record-Keeping and Documentation Procedures
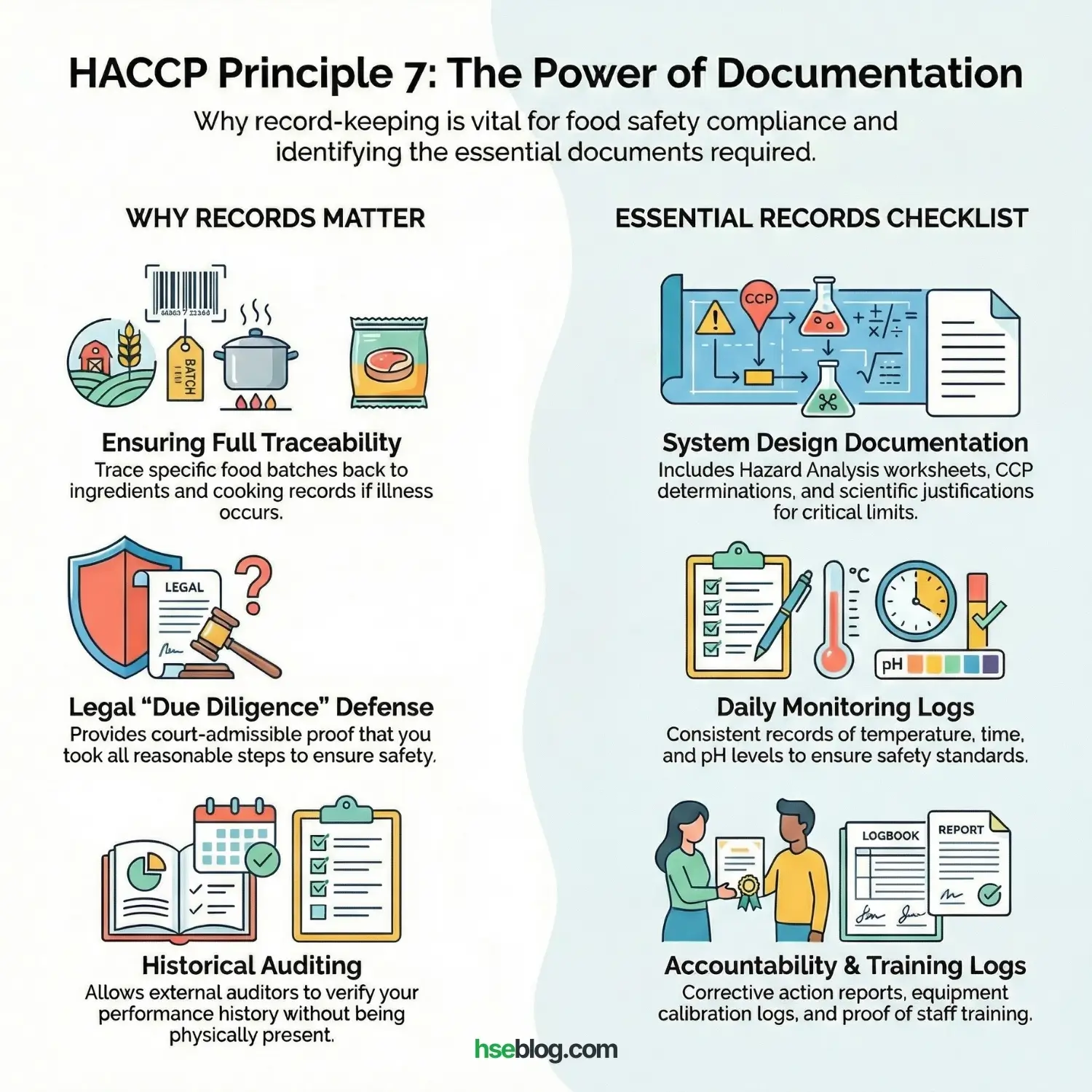
In the HSE and Quality world, “If it isn’t written down, it didn’t happen.” Efficient and accurate record-keeping is essential for the application of the HACCP system.
Why Records Matter:
- Traceability: If a customer gets sick, you can trace the specific batch of food back to the ingredients and cooking records.
- Legal Defense: In court, these records are your proof of “Due Diligence.” They prove you took all reasonable steps to ensure safety.
- Auditing: An external auditor (like me) cannot sit in your kitchen for 365 days. I rely on your records to see the history of your performance.
Essential Records:
- Hazard Analysis Worksheet.
- CCP Determination.
- Critical Limit justification (scientific papers or regulations).
- Daily Monitoring Logs (Temperature, Time, pH).
- Corrective Action Reports.
- Calibration Logs.
- Training Records (proving staff knows how to use the system).
Effective Process For Implementing HACCP Principles
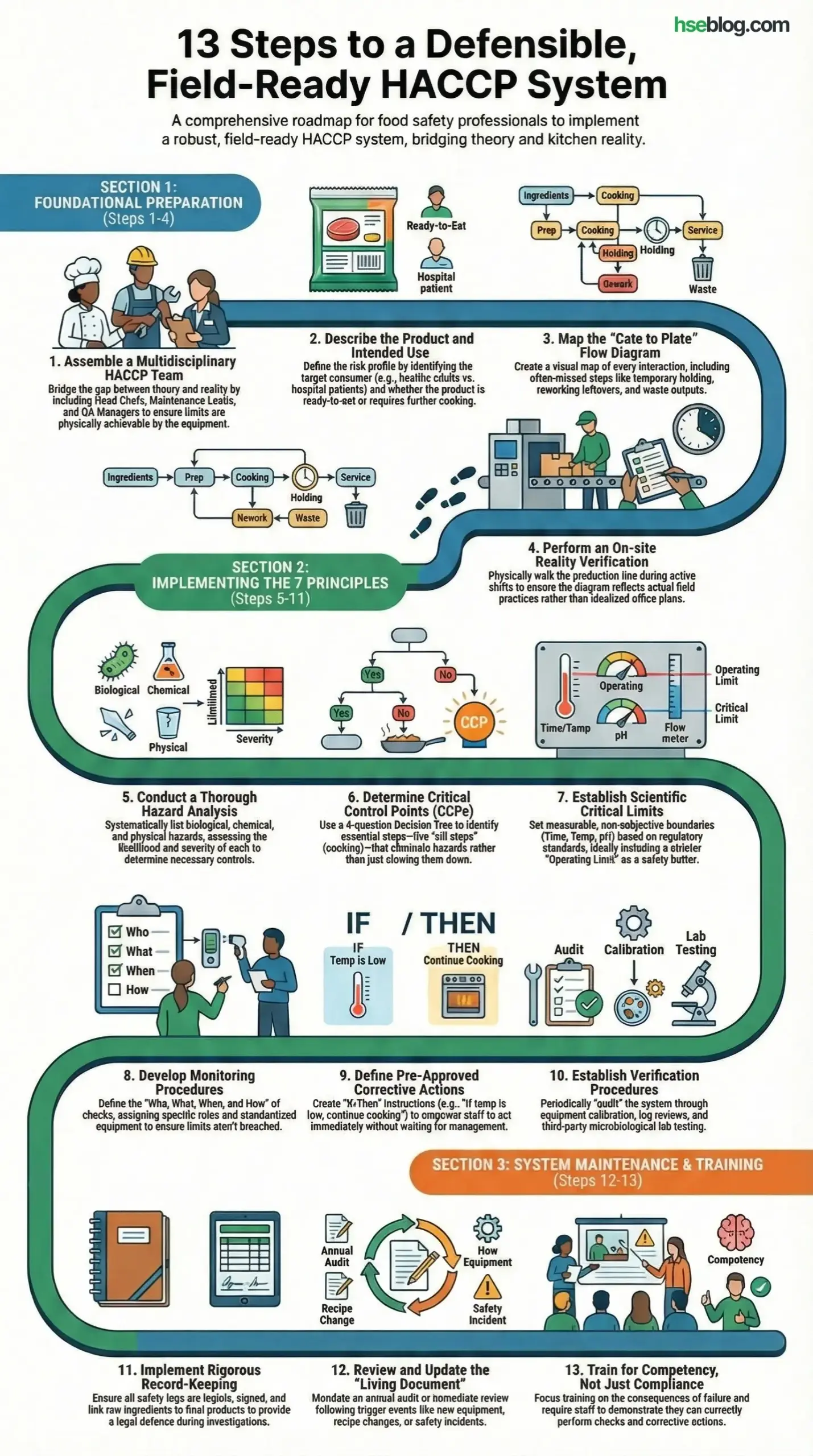
Implementing HACCP is a structured operation. I have found that shortcuts here inevitably lead to critical failures later. Follow these 13 steps to build a defensible, field-ready safety system.
1. Formation of a Multidisciplinary HACCP Team
You cannot write a valid HACCP plan in an office vacuum. I always assemble a team that bridges the gap between theory and reality. This team must include the Head Chef (who knows the recipes), the Maintenance Lead (who knows the freezer performance), and the HSE/QA Manager (who knows the biological risks). Without the maintenance team, for example, you might set a critical limit your equipment is physically incapable of holding.
- Diverse Expertise: Include members from production, sanitation, maintenance, and quality assurance.
- Defined Responsibilities: Clearly assign who writes the plan, who validates it, and who executes it on the floor.
- Resource Allocation: Ensure the team has the authority to request budget for necessary repairs or equipment upgrades.
2. Describe the Product and Intended Use
We must define exactly what we are making and, crucially, who is eating it. In my experience, the risk profile changes drastically depending on the consumer. Providing a raw steak tartare to a healthy oil rig crew is a calculated risk; providing that same dish to a hospital ward or an elderly care facility is negligence. You must also define if the product is ready-to-eat (RTE) or requires further cooking by the consumer.
- Target Consumer: Identify if vulnerable groups (infants, elderly, immunocompromised) are the end-users.
- Product Characteristics: Define shelf life, storage temperature (frozen/chilled/ambient), and packaging type.
- Usage Instructions: Clearly state if the product must be cooked, washed, or diluted before consumption.
3. Develop a Flow Diagram of the Production Process
This is a visual map of the food’s journey from “gate to plate.” I require my teams to map every single interaction the product has, including receiving, storage, thawing, cutting, cooking, cooling, and reheating. Missing a step like “temporary holding” or “reworking leftovers” creates a blind spot where bacteria can multiply unnoticed.
- Comprehensive Mapping: Include all inputs (water, packaging, ingredients) and outputs (waste, rework, finished product).
- Sequence Accuracy: Ensure the diagram follows the exact chronological order of operations.
- Rework Loops: Clearly identify where “failed” product goes—is it trashed or re-cooked?
4. On-site Confirmation of Flow Diagram
Paper plans rarely match field reality. I walk the production line with the flow diagram in hand during active shifts to verify accuracy. I often find that night shifts take shortcuts that the day shift management doesn’t know about, or that product is moved through a “dirty” waste corridor to save time. The diagram must be updated to reflect these actual practices before you can assess the risk.
- Walk the Line: Physically trace the product path during active production hours (including night shifts).
- Verify Inputs/Outputs: Confirm that waste removal paths do not cross clean food paths (cross-contamination).
- Consult Operators: Ask the line workers, “Do you actually do it this way?” to uncover hidden procedural deviations.
5. Conduct a Thorough Hazard Analysis (Principle 1)
Using the verified flow diagram, we systematically list every biological, chemical, and physical hazard that could reasonably occur at each step. In the field, I differentiate between a “quality issue” (stale bread) and a “safety hazard” (moldy bread with mycotoxins). We assess the likelihood of the hazard occurring and the severity of the consequences to determine if it requires control.
- Hazard Identification: List specific agents (e.g., Salmonella, glass shards, cleaning chemicals).
- Risk Assessment: Evaluate Likelihood vs. Severity for each hazard.
- Control Measures: Identify what preventative measures (like sieves, temperature control, or supplier contracts) can control each hazard.
6. Determine the Critical Control Points (Principle 2)
Not every step is a Critical Control Point (CCP). We use a “CCP Decision Tree” to filter the list. A CCP is a step where a control can be applied that is essential to prevent or eliminate the hazard. For example, cold storage prevents growth (Control Point), but cooking kills the bacteria (Critical Control Point). If you have too many CCPs, you will overwhelm your staff; if you have too few, you are taking risks.
- Use a Decision Tree: Apply the 4-question logic model to objectively determine if a step is a CCP.
- Distinguish CP vs. CCP: Separate “Good Manufacturing Practices” (hand washing, general cleaning) from “Critical Controls” (pasteurization, metal detection).
- Focus on Kill Steps: Prioritize steps that eliminate the hazard rather than just slowing it down.
7. Establish Critical Limits for Each CCP (Principle 3)
For every CCP, we must set a hard, scientific boundary between “safe” and “unsafe.” These are not opinions; they are based on regulatory standards (FDA, local HSE laws) or scientific data. I always advise setting an “Operating Limit” that is stricter than the “Critical Limit.” If the Critical Limit is 75°C, we aim for 77°C. This buffer allows operators to intervene before the legal limit is breached.
- Measurable Parameters: Limits must be quantifiable (Time, Temperature, pH, Moisture Level), not subjective.
- Regulatory Alignment: Ensure limits meet or exceed local legal food safety requirements.
- Validation: Ensure the chosen limit is scientifically proven to control the specific hazard (e.g., Salmonella death curves).
8. Develop Monitoring Procedures (Principle 4)
We must define the “Who, What, When, and How” of monitoring. I prefer continuous monitoring (like automated digital recorders) where possible, but manual checks are often necessary. The frequency of monitoring must be sufficient to ensure that the Critical Limit has not been breached for a long period. If you check a fridge once a day, and it fails 10 minutes after the check, you lose 24 hours of product.
- Define Responsibility: Assign a specific role (e.g., “Grill Cook”) to the task, not a generic department.
- Set Frequency: Determine how often checks occur (e.g., “Every 30 minutes” or “Every batch”).
- Standardize Methods: Provide specific equipment (calibrated probes) and instructions on how to use them.
9. Define Corrective Actions (Principle 5)
When a critical limit is breached, the operator needs a pre-approved action plan. They should not have to call a manager to ask what to do while the food is spoiling. I write these actions as “If/Then” statements. For example: “If the internal temp is <74°C, continue cooking. If it cannot reach temp, discard immediately.” This empowers the workforce to protect safety without fear of reprimand for waste.
- Immediate Action: Instructions to bring the process back under control (e.g., “Stop the line,” “Re-heat”).
- Product Disposition: Clear rules on what to do with the affected product (Hold, Test, or Destroy).
- Root Cause Analysis: A mechanism to investigate why the deviation happened to prevent recurrence.
10. Establish Verification Procedures (Principle 6)
Verification confirms that the system is working as intended. This is the “audit” phase. While monitoring checks the product, verification checks the process. This includes calibrating thermometers, reviewing the daily logs for falsification or errors, and sending random samples to a third-party lab to prove that our cooking times are actually killing the bacteria.
- Calibration: regularly checking monitoring tools against a known standard to ensure accuracy.
- Records Review: A weekly management review of CCP logs to ensure checks were actually performed.
- Microbiological Testing: Periodic lab testing of the final product to validate the entire HACCP plan.
11. Implement Record-keeping and Documentation Systems (Principle 7)
In the eyes of a regulator or a court, if it isn’t documented, it didn’t happen. I insist on rigorous documentation because it is our only defense in an investigation. Records must be legible, signed, dated, and unalterable. Digital systems are preferred now, but paper logs must be kept in waterproof binders in wet operational areas.
- Traceability: Records must link raw ingredients to the final product (Batch tracking).
- Accessibility: Logs must be readily available for inspection by auditors or health authorities.
- Retention: Keep records for the appropriate duration based on product shelf life and legal requirements (often 1-2 years).
12. Review and Update the HACCP Plan Regularly
A HACCP plan is a living document, not a “set and forget” file. I mandate a review of the plan at least annually, or immediately whenever there is a significant change—such as a new raw material supplier, a new piece of equipment, a recipe change, or an actual safety incident. This ensures the plan evolves with the operation.
- Trigger Events: Define what changes trigger a review (e.g., new menu items, new packaging).
- Annual Validation: Conduct a full system audit once a year even if nothing has changed.
- Incident-Based Review: Re-evaluate the hazard analysis immediately following any near-miss or recall.
13. Train and Educate Staff
The most perfect paperwork cannot save you if the cook doesn’t understand why the temperature matters. I focus training on the consequences of failure—showing staff that their actions directly prevent people from getting sick. Competency is key; it is not enough to show them how to use a thermometer; they must demonstrate they can do it correctly and know the corrective actions.
- Role-Specific Training: Tailor training to the specific CCPs the employee is responsible for.
- Refresher Training: Regular updates to keep safety top-of-mind and address bad habits.
- Competency Assessment: Verify learning through practical observation, not just a written quiz.
Conclusion
HACCP is the difference between hoping food is safe and knowing it is safe. In my career, I have seen that the sites with robust HACCP plans run smoother, have less waste, and maintain higher morale because the workforce trusts the food they are eating.
Effective implementation requires discipline. It requires managers who are willing to throw away expensive product because a critical limit was breached. That is the cost of safety. As safety leaders, our job is to enforce these principles rigorously, ensuring that production pressure never overrides the critical controls designed to protect human health.

