During an industrial hygiene audit at a university research facility, I walked into a chemistry lab to find a post-doctoral researcher handling concentrated sulfuric acid while wearing shorts, sandals, and standard prescription glasses. I immediately halted the procedure, stepping in before a minor slip could turn into a life-altering chemical burn. Academic brilliance does not make anyone immune to the laws of chemistry and physics, yet I frequently see basic safety principles casually ignored in research environments.
Chemical splashes, toxic fumes, thermal burns, and biological exposures do not discriminate between a first-year student and a senior principal investigator. This article covers the ten essential pieces of Personal Protective Equipment (PPE) every student and researcher must use, detailing how they function in real operations and why they are critical. When you step into a laboratory, PPE is not a bureaucratic requirement; it is the physical barrier keeping hazardous materials from permanently altering your life.
TL;DR
- PPE is the final line of defense in the Hierarchy of Controls; if a chemical reaches your PPE, a primary safety system has already failed.
- Standard prescription glasses offer zero splash protection; proper chemical-splash goggles are non-negotiable in the lab.
- Never wear laboratory gloves in public spaces like hallways or elevators to prevent hazardous cross-contamination.
- Inspect your gear before every use; degraded or damaged PPE creates a dangerous illusion of safety.
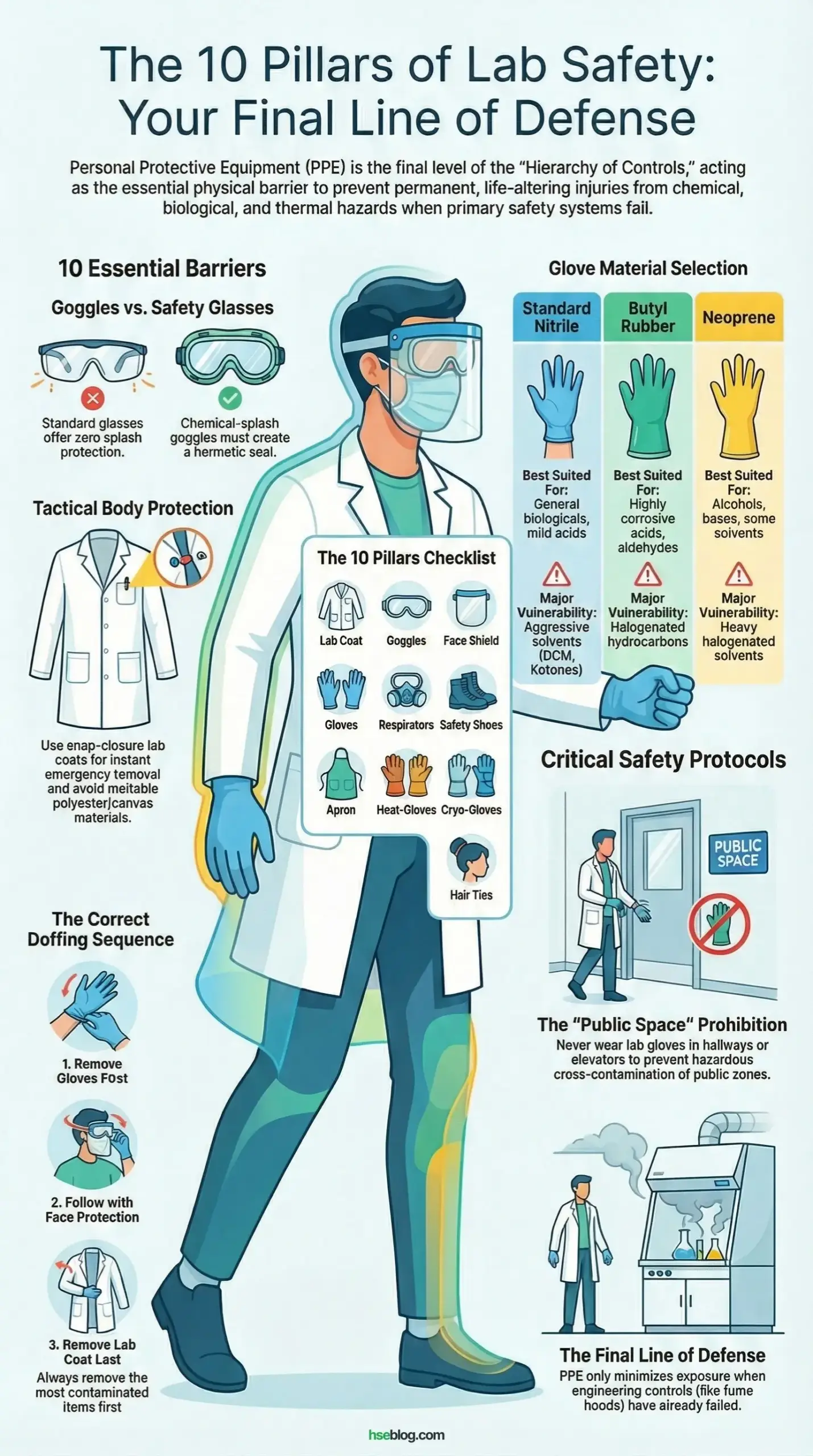
What Is PPE in the Laboratory?
Personal Protective Equipment (PPE) encompasses specialized clothing and gear designed to protect personnel from chemical, biological, physical, and mechanical hazards. In the laboratory, this equipment serves as a critical, life-saving barrier between the human body and volatile reactions. It is vital to understand that PPE does not eliminate the hazard itself; it merely minimizes exposure when all other safety systems fail.
As an Industrial Hygienist, I always enforce the Hierarchy of Controls on any site. We first try to eliminate the hazard, substitute it, use engineering controls like fume hoods, and implement administrative controls. PPE is the absolute last line of defense. If a highly corrosive chemical is eating through your nitrile glove, it means the primary containment failed, and that thin layer of rubber is the only thing saving your skin.
Why PPE Is Critical for Students and Researchers
Academic and research laboratories often handle a wider, more unpredictable variety of hazardous chemicals than standardized industrial manufacturing plants. Students and researchers face daily risks from unexpected exothermic reactions, pressurized glassware failures, and biological contamination. The variable nature of research means hazards can change from one bench to the next.
School labs usually deal with diluted solutions, but advanced research labs frequently handle concentrated acids, pyrophoric materials, and unknown synthesized compounds. Institutional safety requirements and legal standards, like OSHA’s Laboratory Standard (29 CFR 1910.1450), mandate strict PPE compliance. Ignoring these rules does not just risk regulatory fines; it risks irreversible physical trauma, blindness, or severe occupational illness.
The 10 Essential PPE Items for Laboratory Operations
Every laboratory environment requires a specific matrix of protective equipment based on a rigorous, documented risk assessment. Standardizing basic PPE ensures that no matter what experiment is running, a baseline of bodily protection is rigidly maintained. Below are the ten non-negotiable items every researcher must utilize when exposed to lab hazards.
1. The Laboratory Coat: Your Primary Dermal Barrier

The lab coat is engineered to take the initial kinetic impact and chemical load of a splash, buying you critical seconds to react and strip the garment. It is not a fashion statement or a temperature control layer; it is a tactical, consumable barrier.
Open coats provide zero frontal protection and act as a funnel, directing spilled chemicals straight onto your street clothes and skin. You must evaluate the hazard before selecting the material, as standard cotton blends ignite easily. Polyester blends are even worse; they melt directly into the skin during a flash fire, drastically complicating emergency trauma care.
- Fully button or snap the coat from top to bottom before initiating any task.
- Use flame-resistant (FR) Nomex coats when handling pyrophorics or bulk flammables.
- Ensure the sleeves completely overlap your glove cuffs to prevent liquid channeling down your arms.
Pro Tip: Always choose lab coats with snap closures over traditional buttons. In a severe chemical saturation emergency, you must be able to rip the garment off in a single, violent motion without fumbling.
2. Chemical-Splash Goggles: Non-Negotiable Eye Protection

Goggles must create a hermetic or heavily restricted rubber seal around your entire orbital bone structure. A chemical splash does not travel in a predictable straight line; it deflects off benchtops, atomizes, and searches for the path of least resistance.
The most dangerous habit I constantly correct during lab inspections is the use of standard safety glasses during liquid transfers. Liquids will easily bypass the massive gaps at the brow and temples of standard glasses, channeling directly into the eyes. You must utilize indirect-vented or non-vented goggles for volatile chemicals.
- Discard heavily scratched goggles immediately; compromised vision leads to fatal procedural errors.
- Invest in premium anti-fog coatings to prevent the dangerous reflex of removing eyewear mid-task to wipe them.
- Verify the rubber seal makes uninterrupted, tight contact with your face before handling any reagent.
3. Face Shields: Ballistic and Secondary Deflectors

A face shield is a macro-level physical barrier designed to absorb high-energy impacts and massive fluid displacement. It is deployed when the hazard volume or kinetic pressure significantly exceeds the capacity of standard facial PPE.
A face shield is strictly secondary protection and must never be worn without chemical-splash goggles underneath. I have investigated severe eye injuries where an upward-deflecting splash traveled directly under a face shield’s visor into unprotected eyes. It is your blast shield during high-risk pressurized or exothermic reactions.
- Deploy during the bulk decanting of concentrated corrosives or reactive alkali metals.
- Use as a mandatory thermal and splash barrier against cryogenic fluid expansion.
- Rely on the heavy polycarbonate visor to absorb glass shrapnel from imploding vacuum flasks.
4. Disposable Gloves: Understanding Chemical Permeation

Disposable gloves are a temporary, highly specific dermal barrier governed by complex chemical degradation and permeation rates. There is absolutely no such thing as a “universal” glove in a chemical laboratory environment.
Standard 4-mil nitrile gloves provide excellent general biological resistance but will fail in seconds against aggressive solvents like dichloromethane or concentrated nitric acid. Once a chemical permeates the material, the glove acts as an occlusive dressing, trapping the toxin against your skin and accelerating chemical burns.
| Glove Material | Best Suited For | Major Vulnerability |
| Standard Nitrile | General biologicals, mild acids, incidental contact | Aggressive solvents (ketones, DCM) |
| Butyl Rubber | Highly corrosive acids, aldehydes, ketones | Aliphatic and halogenated hydrocarbons |
| Neoprene | Alcohols, bases, some solvents | Heavy halogenated solvents |
- Consult a chemical glove compatibility chart before starting any new reaction or solvent transfer.
- Change gloves immediately upon suspected chemical contact, visible degradation, or micro-tears.
- Execute the strict “glove-in-glove” peeling technique to avoid transferring surface toxins to bare hands during removal.
5. Respiratory Protection: The Final Filtration Line

Respirators are deployed strictly when primary engineering controls like laboratory fume hoods are bypassed, unavailable, or actively failing. Handing an untested respirator to a researcher is a massive operational liability that provides a lethal illusion of safety.
Surgical masks offer zero filtration against chemical vapors or hazardous particulates and have no place in chemical safety. Depending on the risk, you need an N95 for fine powders or an elastomeric half-mask with highly specific chemical cartridges. Even a single day’s facial hair growth will break the tight-fitting seal, rendering the respirator completely useless against toxic fumes.
- Mandate quantitative fit testing and medical clearance before assigning any tight-fitting respirator.
- Never cross-apply cartridges; an organic vapor cartridge cannot and will not filter out acid gases.
- Implement a strict, mathematically calculated replacement schedule for all chemical cartridges based on exposure limits.
“A respirator that does not seal perfectly is worse than no respirator at all; it mathematically encourages a worker to confidently step into a fatal atmosphere.”
6. Safety Shoes and Closed-Toe Footwear

Gravity ensures that every spilled chemical and dropped piece of heavy equipment will eventually strike the floor. In a laboratory environment, your footwear is not a matter of ergonomic comfort; it is your baseline structural defense against liquid saturation and blunt force impact.
I routinely eject personnel from labs for wearing canvas sneakers, mesh running shoes, or ballet flats. These porous materials offer zero physical protection and actively worsen chemical burns by instantly absorbing corrosive liquids, acting as a wick that traps the hazard directly against your skin. A proper lab shoe must be completely enclosed from heel to toe, constructed of a non-porous material like solid leather or dense synthetics.
In scaled-up research labs, pilot plants, or when personnel are handling compressed gas cylinders, standard closed-toe shoes are completely inadequate. Operations in these heavy environments require an immediate upgrade to chemical-resistant safety boots with steel or composite toe caps.
- Ban all porous footwear on the lab floor, including canvas slip-ons and perforated clogs.
- Ensure all shoe soles are heavily slip-resistant to safely navigate slick, wet chemistry environments.
- Mandate safety-toe boots for any personnel moving heavy chemical drums or operating pilot machinery.
7. Chemical-Resistant Apron

A chemical-resistant apron provides a heavy-duty, impermeable macro-barrier engineered specifically for high-risk, high-volume material transfers. When you scale up a reaction, a standard cloth lab coat simply lacks the absorption capacity or chemical resistance to protect your vital organs from a massive breach.
I strictly mandate thick rubber, neoprene, or PVC aprons whenever a worker is decanting large volumes of concentrated acids, caustics, or highly toxic liquids. If a runaway exothermic reaction causes a mixture to boil over, the apron takes the violent brunt of the corrosive attack. This impermeable shield buys you the critical physiological time required to step back and reach the emergency safety shower.
“A lab coat absorbs; a chemical apron deflects. Never confuse the two when handling bulk corrosives.”
Pro Tip: Always cross the apron strings behind your back and tie them loosely in the front. In a severe chemical saturation emergency, you must be able to pull a single slip-knot and violently shed the contaminated apron instantly, rather than fumbling blindly behind your back.
8. Heat-Resistant Gloves

Heat-resistant gloves are essential, specialized barriers for handling materials removed from autoclaves, muffle furnaces, or standard hot plates. Attempting to use generic lab gloves for extreme thermal tasks is a guaranteed path to severe, life-altering combination burns.
Students and rushed researchers often make the critical mistake of using standard nitrile or latex gloves to move hot glassware. Upon contact with extreme heat, these thin synthetic materials instantly melt directly into the human epidermis, requiring extensive surgical debridement to remove.
Proper thermal gloves are typically constructed from heavy leather, Kevlar, or Zetex. However, workers must understand a massive vulnerability: these woven materials are generally not waterproof or chemical-resistant. If a hot, corrosive liquid spills onto a woven heat-resistant glove, the liquid will immediately soak through the fabric and burn the worker’s skin.
9. Cryogenic Gloves

Cryogenic gloves are heavily insulated, specialized PPE designed strictly to prevent severe tissue frostbite when handling ultra-low temperature materials like liquid nitrogen or dry ice. They allow for the safe, tactile manipulation of frozen sample vials and pressurized cryogenic valves.
It is a vital, non-negotiable operational rule that cryogenic gloves are designed solely for incidental contact and splash protection. They are never engineered for total liquid immersion.
If a worker incorrectly assumes the glove is impenetrable and submerges their hand directly into a dewar of liquid nitrogen, the ultra-cold liquid will instantly seep through the seams. The liquid will then rapidly freeze the insulated glove directly to the worker’s hand, causing catastrophic tissue damage and severe deep-layer frostbite.
10. Hair Protection (Ties and Caps)

Loose, long hair is a constantly overlooked mechanical and flammability hazard on the laboratory floor. It easily acts as a wick for chemical contamination, a rapid fuel source for open flames, and a capture point for rotating machinery.
Securing hair is a fundamental, non-negotiable safety requirement, not a cosmetic suggestion or an arbitrary administrative rule. Hair easily catches fire from unguarded Bunsen burners or drapes directly into highly contaminated chemical mixtures when a researcher leans over a benchtop.
- Tie hair back tightly and securely above the collar line before crossing the laboratory threshold.
- Utilize disposable bouffant caps in sterile environments, microbiology labs, or clean rooms.
- Never operate a rotary evaporator, centrifuge, or lathe with unsecured hair or dangling lanyards.
I have investigated a severe incident where a researcher’s loose hair contacted a rotating centrifuge shaft. The kinetic energy of the machine instantly entangled the hair, resulting in a severe, traumatic scalp laceration before the emergency stop could be hit.
How to Properly Wear and Remove PPE
Proper donning (putting on) and doffing (taking off) of PPE is just as important as the equipment itself. If you remove contaminated gear haphazardly, you will transfer hazardous chemicals directly onto your bare hands, face, or clothing. The sequence matters immensely, especially when dealing with high-toxicity materials.
When donning, generally start with the lab coat, followed by eye protection, and put gloves on last, ensuring they pull tightly over the cuffs of the coat. When doffing, remove the most contaminated items first. Gloves should come off first, followed by the face shield, goggles, and finally the lab coat, washing hands immediately and thoroughly afterward.
Common Mistakes Students Make with PPE
The most persistent failure I observe in academic environments is the migration of lab PPE into public spaces. Students frequently wear contaminated gloves while opening doors, pressing elevator buttons, or handling mobile phones. This cross-contaminates safe zones, exposing unprotected janitorial staff and fellow students to severe chemical hazards.
“Wearing PPE outside the lab does not make you look like a dedicated scientist; it marks you as a direct hazard to public health.”
Another common error is reusing single-use disposable PPE to save money or time. A nitrile glove that has been exposed to a solvent may look intact but has likely lost its chemical resistance on a molecular level. Similarly, failing to fasten lab coats or wearing goggles resting on the forehead defeats the entire engineering of the protective equipment.
Maintenance and Replacement of PPE
PPE is subjected to harsh conditions and will degrade over time; it requires routine visual inspection before every single use. Check lab coats for chemical stains or fraying, inspect goggles for deep scratches that impair vision, and stretch gloves slightly to check for micro-tears. Operating with damaged PPE provides zero protection while giving the worker a false, dangerous sense of security.
Reusable PPE must be cleaned according to the manufacturer’s strict specifications. Face shields and goggles should be washed with mild soap and water, not harsh lab solvents that degrade the protective plastic. If a lab coat becomes heavily contaminated with a hazardous substance, it must be disposed of as hazardous waste, not casually thrown into a standard washing machine.
Conclusion
Working in a laboratory is a privilege that comes with the profound, daily responsibility of managing life-threatening hazards. PPE is not an administrative hurdle designed to slow down your research; it is the final physical barrier standing between a routine task and a permanent disability. Academic pressure and publication deadlines must never override fundamental safety protocols.
Building a proactive safety culture starts with the individual choices you make at the bench. Consistently wearing and maintaining your PPE builds a habit that protects not only you but your colleagues. Ultimately, the rule in any high-risk environment is non-negotiable: No PPE, no experiment. Human life and physical well-being will always take absolute priority over scientific curiosity.

