TL;DR — Key Numbers
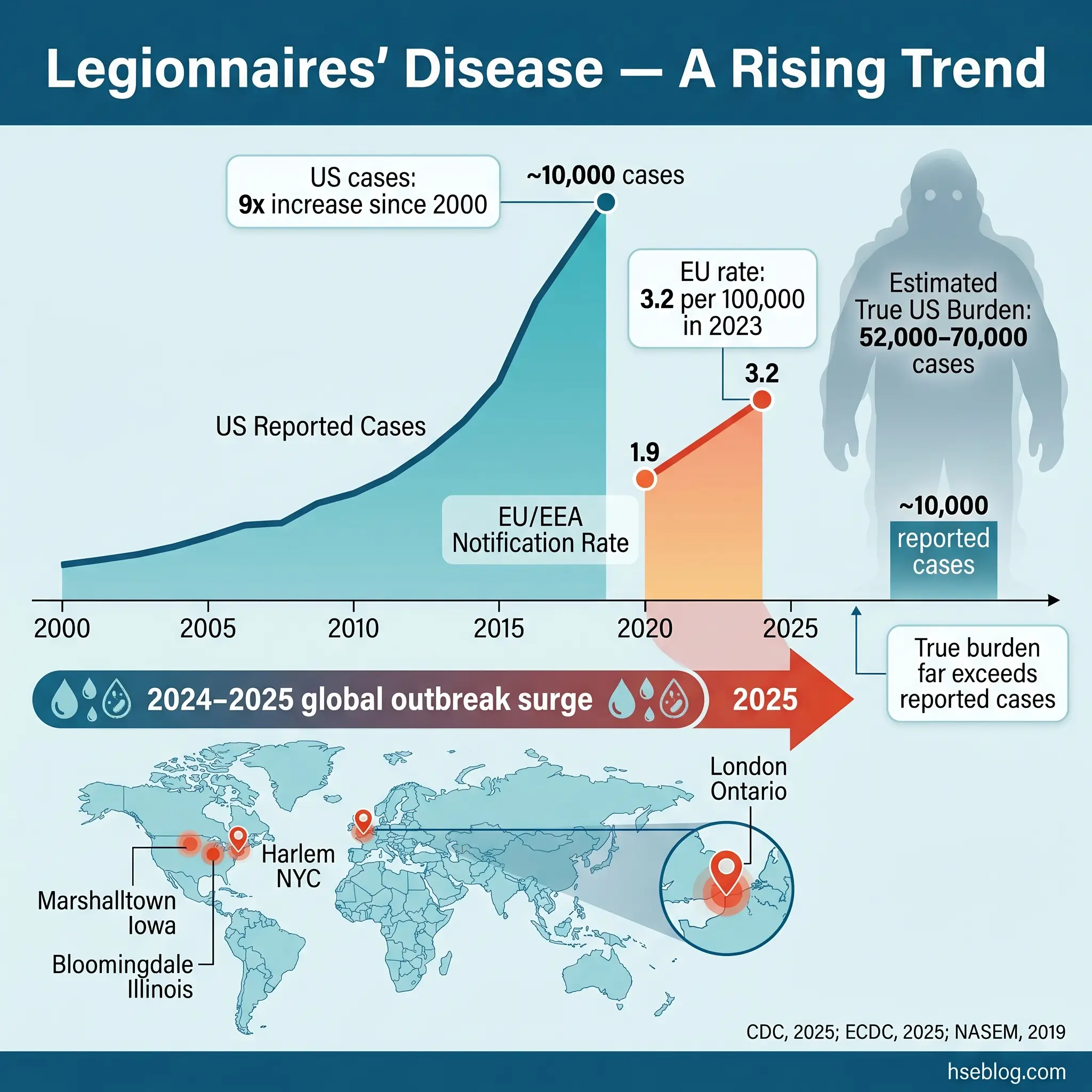
- 52,000–70,000 estimated actual annual US Legionnaires’ disease cases versus ~10,000 reported — most cases are never diagnosed (National Academies of Sciences, Engineering, and Medicine, 2019).
- 5–10% overall case-fatality rate; rises to 40–80% in untreated immunocompromised patients (WHO, 2022).
- 20–45°C (68–113°F) — the water temperature window where Legionella proliferates; every building water system operating in this range without active control is amplifying risk (CDC, 2025).
- 3.2 per 100,000 — EU/EEA Legionnaires’ disease notification rate in 2023, the highest ever recorded (ECDC, 2025).
Legionella is a genus of waterborne bacteria — most notably Legionella pneumophila — that causes Legionnaires’ disease, a severe form of pneumonia, and the milder Pontiac fever. Naturally present at low levels in freshwater, Legionella becomes a health hazard when it amplifies in engineered water systems between 20°C and 45°C and is inhaled as contaminated aerosols. Prevention depends on temperature control, stagnation elimination, and a documented water management program led by a competent responsible person.
What Is Legionella?
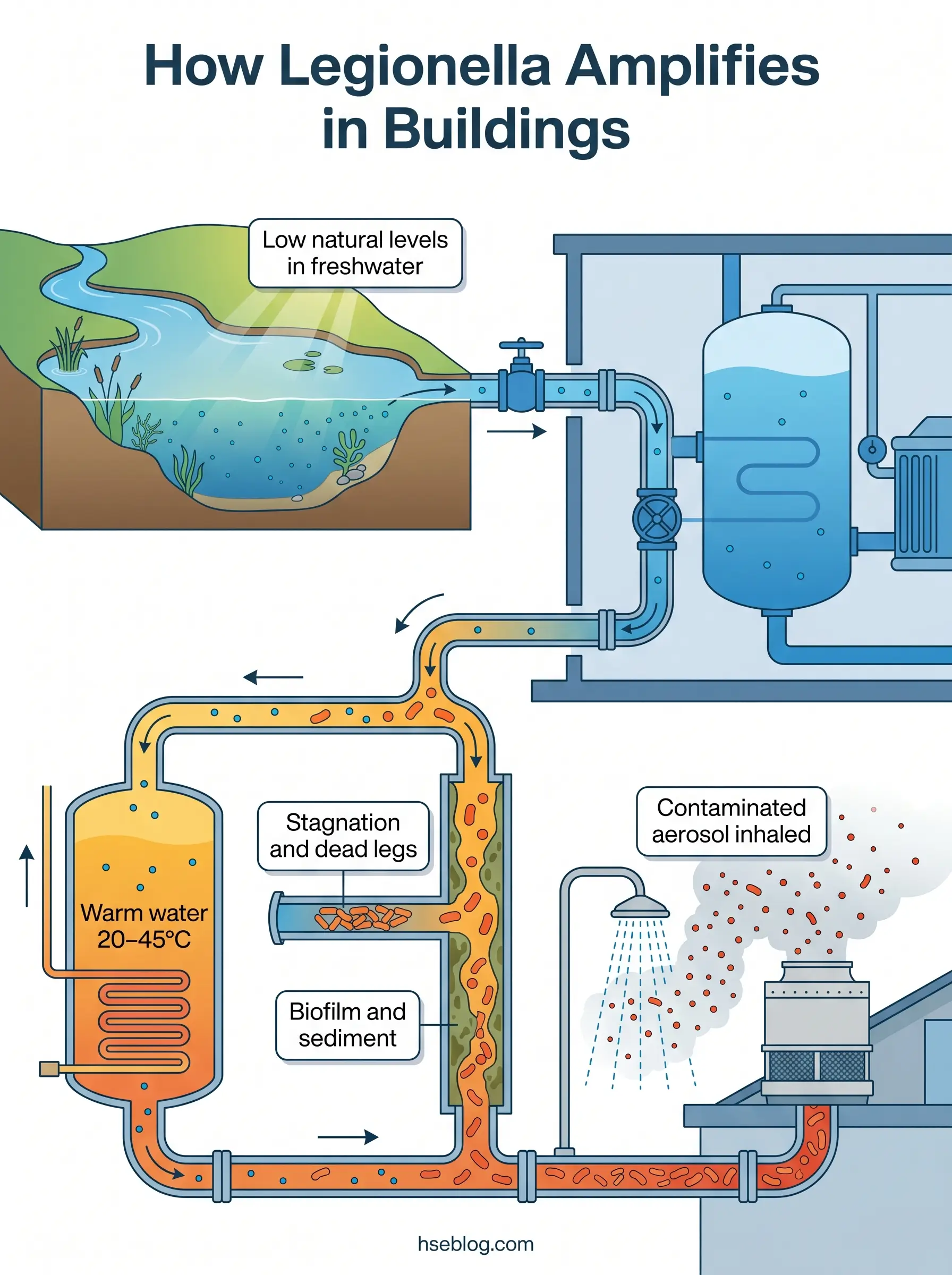
Legionella is a genus of gram-negative bacteria with more than 60 identified species. The species responsible for the vast majority of human illness is Legionella pneumophila, and within that species, serogroup 1 accounts for roughly 90% of culture-confirmed clinical cases. The bacteria exist naturally in lakes, rivers, and soil at concentrations too low to cause harm. The problem begins when Legionella enters engineered water systems — cooling towers, hot water storage vessels, distribution pipework — and finds conditions that let it multiply.
Those conditions are well characterised. Legionella proliferates in water between 20°C and 45°C (68–113°F), with the most rapid growth occurring between 25°C and 45°C (CDC, 2025). Add stagnation, biofilm on pipe surfaces, sediment, and low or absent disinfectant residual, and a system that was microbiologically unremarkable at commissioning can become a significant amplification site within weeks.
The single biggest public misconception I encounter in HSE practice is the belief that Legionella “comes from the water supply.” In almost every documented outbreak, the contamination is an in-building amplification problem. Municipal water treatment delivers water with residual disinfectant and at temperatures below the growth-favourable range. What happens inside the building — how water is stored, heated, circulated, and used — determines whether Legionella reaches dangerous concentrations. This is precisely why legal responsibility sits with building owners and duty holders, not water utilities.
Legionnaires’ Disease vs. Pontiac Fever: The Two Forms of Legionellosis
A consistent source of confusion — among workers, facility managers, and sometimes clinicians — is the relationship between Legionnaires’ disease and Pontiac fever. Both are caused by Legionella bacteria, both result from inhaling contaminated aerosols, but they are clinically very different conditions. The umbrella term covering both is “legionellosis.”
Legionnaires’ disease is the severe form: a pneumonia with an incubation period of 2–10 days (median 5–6 days), requiring antibiotic treatment, and carrying a case-fatality rate of 5–10% overall (WHO, 2022). Some outbreak investigations have documented incubation periods extending to 16–19 days (CDC, 2025; Robert Koch Institute LeTriWa study, 2023). Pontiac fever is the self-limiting, non-pneumonic form: flu-like symptoms appearing within 24–48 hours (range 5–66 hours), resolving without antibiotics in 2–5 days.
The attack-rate difference is striking and operationally important. In an exposed group, Pontiac fever can affect up to 90% of those exposed, while Legionnaires’ disease typically develops in fewer than 5%. Many reported “suspected Legionella” workplace clusters that I have reviewed in HSE practice turn out to be Pontiac fever — the high attack rate creates alarm, but the clinical course is far milder, and the investigation posture shifts from urgent clinical management toward source identification and control.
A rare third category — extrapulmonary legionellosis — can affect sites outside the lungs (heart, wounds, brain) in severely immunocompromised patients, but this is uncommon and typically healthcare-associated.
| Feature | Legionnaires’ Disease | Pontiac Fever |
|---|---|---|
| Type | Pneumonic | Non-pneumonic |
| Incubation | 2–10 days (up to 16–19) | 24–48 hours (range 5–66 hrs) |
| Key symptoms | High fever, cough, shortness of breath, chest pain, GI symptoms, confusion | Fever, muscle aches, headache |
| Severity | Potentially fatal pneumonia | Self-limiting flu-like illness |
| Treatment | Antibiotics required | No antibiotics needed |
| Attack rate | ~5% of exposed | Up to ~90% of exposed |
| Typical outcome | Hospitalisation; ICU in ~1/3 of cases | Full recovery in 2–5 days |
Where Does Legionella Grow? Common Sources in Buildings
Understanding where Legionella amplifies is the foundation of every water management program. The bacteria require the same four conditions regardless of the system type: temperature in the growth-favourable range, stagnation, a nutrient source (biofilm, sediment, scale), and insufficient disinfectant residual. What varies is which building systems provide those conditions most readily.
The highest-consequence source historically is the cooling tower and evaporative condenser. These systems aerosolise water as part of their design function, and drift from contaminated cooling towers has been traced to cases more than 1.5 kilometres from the source. The 2025 Harlem (NYC) community outbreak — 114 cases and 7 deaths, with twelve cooling towers testing positive (NYC Department of Health, 2025) — is a recent example of this transmission pattern.
Other common amplification sites include:
- Hot and cold water systems in large or complex buildings — calorifiers (hot water storage cylinders), thermostatic mixing valves (TMVs), dead legs (sections of pipework with no regular flow), and long distribution runs where temperature drops below control thresholds
- Whirlpool spas and hot tubs — particularly in hotels, leisure centres, and cruise ships, where warm, aerated water is the operating design
- Decorative fountains and misting systems — any feature that generates fine water droplets in a public area
- Respiratory therapy equipment, CPAP devices, and ice machines in healthcare — where even small-volume water reservoirs become significant because patients are already in high-risk groups
- Less-obvious sources — vehicle windshield-washer reservoirs, potting compost (L. longbeachae, particularly documented in Australia and New Zealand), and emergency eyewash/shower stations that are installed but rarely flushed
Watch For: The high-consequence failure pattern in the published outbreak record is not a system that was never controlled — it is a system where control lapsed during a known transition. Building re-occupation after extended closure, seasonal shutdown of one cooling-tower cell, TMV retrofit without post-installation risk review, or prolonged tenant vacancy all create the stagnation-plus-temperature conditions Legionella exploits. Post-COVID building re-openings were a documented contributor to the 2024 global outbreak surge (The Lancet Microbe, 2025).
How Does Legionella Spread to People?
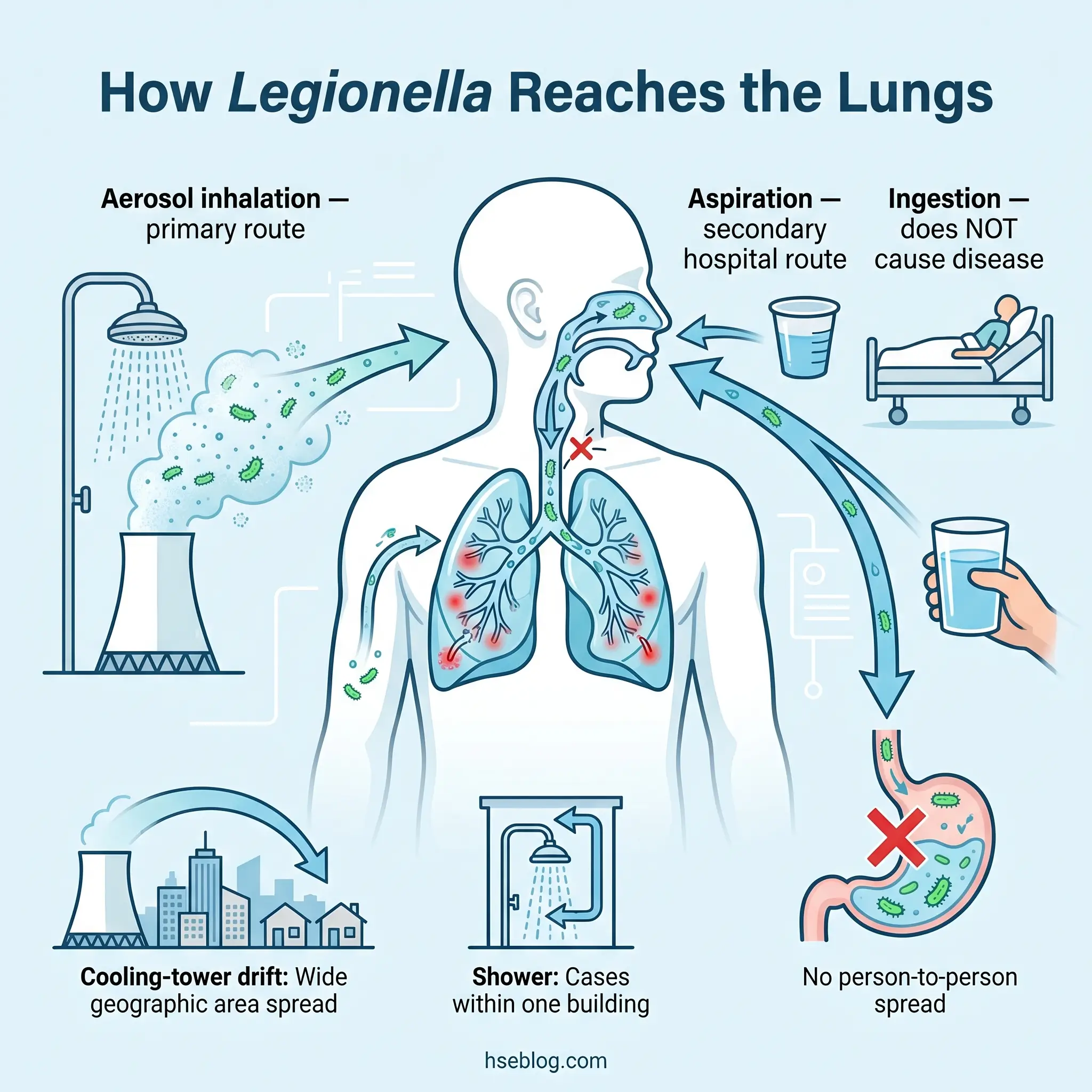
The primary transmission route is inhalation of water aerosols — droplets small enough (typically less than 5 µm) to penetrate deep into the lower respiratory tract. Showers, cooling-tower drift, spa jets, decorative fountains, and misting systems all generate these fine droplets. Drinking contaminated water does not cause Legionnaires’ disease through the digestive tract.
A secondary route, clinically relevant in hospitals, is aspiration — water “going down the wrong pipe” into the lungs rather than the stomach. Patients with swallowing difficulties, those with nasogastric tubes, or those recently extubated are vulnerable to this mechanism, which is why healthcare water-system management carries heightened regulatory scrutiny under both CMS requirements in the US and ACoP L8 in the UK.
Person-to-person transmission is not established. One probable case has been documented globally, but Legionella is fundamentally an environmental pathogen, not a contagious one. This distinction matters operationally: outbreak investigation focuses on identifying the environmental source, not on isolating patients.
The transmission mechanism also explains why different source types produce different outbreak patterns. Cooling-tower outbreaks generate geographically dispersed cases across the wind-drift footprint — cases appearing across multiple postcodes, with no common indoor exposure. Shower-linked or spa-linked outbreaks produce facility-clustered cases — guests at one hotel, residents of one building. Investigators use this geographic pattern as an early signal to narrow the source search.
What Are the Symptoms of Legionnaires’ Disease?
Legionnaires’ disease typically presents 2–10 days after exposure, with a median incubation of 5–6 days (CDC, 2025). Early symptoms are non-specific — headache, muscle aches, fatigue, loss of appetite, and a low-grade fever that can initially resemble influenza.
By days 2–3, the clinical picture sharpens. Symptoms that should raise concern include:
- High fever, often exceeding 39–40°C (102–104°F)
- Cough, initially dry but potentially becoming productive with mucus or, in some cases, blood-tinged sputum
- Shortness of breath and chest pain, reflecting lower-respiratory involvement
- Gastrointestinal symptoms — diarrhoea, nausea, and abdominal pain occur in a significant proportion of cases, which is atypical for most bacterial pneumonias
- Neurological symptoms — confusion, altered mental status, headache beyond what the fever alone would explain
The gastrointestinal and neurological features are the clinical markers that distinguish Legionnaires’ disease from more common community-acquired pneumonias. A triad of high fever, diarrhoea, and confusion in a patient with pneumonia — particularly one over 50 with underlying risk factors — should trigger specific Legionella testing rather than empirical treatment for typical pneumonia organisms alone. As discussed in the WHO Legionellosis fact sheet, case-fatality runs 5–10% overall, rising significantly without appropriate antibiotic therapy.
Pontiac fever presents quite differently: fever, muscle aches, and headache without pneumonia, resolving within 2–5 days. No specific treatment is required.
Field Test: If a building water-system investigation is triggered by reported illness, confirming whether the clinical presentation is Legionnaires’ disease or Pontiac fever changes the urgency and scope of the response. The first question an HSE professional should ask the public-health authority is whether pneumonia has been confirmed.
Who Is Most at Risk of Legionnaires’ Disease?
Most healthy people exposed to Legionella-contaminated aerosols do not develop Legionnaires’ disease. The factors that convert exposure into clinical illness are well documented and cluster into demographic, medical, and behavioural categories.
Age is the strongest single predictor — 75–80% of reported cases occur in people aged 50 and over (WHO, 2022). Male sex is associated with higher case rates, with 60–70% of reported cases occurring in males (WHO, 2022). Current and former smokers carry elevated risk due to compromised mucociliary clearance. Chronic lung disease (COPD, emphysema), diabetes, kidney disease, cancer, and heart failure each independently increase susceptibility.
Immunosuppression — whether from HIV, chemotherapy, transplant anti-rejection medications, or long-term corticosteroid use — dramatically changes the clinical equation. Case-fatality in untreated immunocompromised patients reaches 40–80% (WHO, 2022). Hospitalised patients face additional risk, particularly those who are intubated, have nasogastric tubes, or use respiratory therapy equipment where small-volume water reservoirs may harbour the bacteria.
The judgment call for facility managers in healthcare and residential-care settings is that the “average person’s risk is low” framing breaks down entirely when the building population is concentrated in high-risk groups. Every occupant in a care home, every patient on an immunosuppression ward, should be treated as higher-risk by default. This is why both HSE ACoP L8 (UK) and CMS requirements (US) treat healthcare and care-home water systems with elevated scrutiny — the duty of care scales with the vulnerability of the population served.
How Is Legionnaires’ Disease Diagnosed and Treated?
This section provides general clinical information for HSE practitioner awareness. It is not medical advice. Individuals with symptoms consistent with Legionnaires’ disease should seek immediate medical attention.
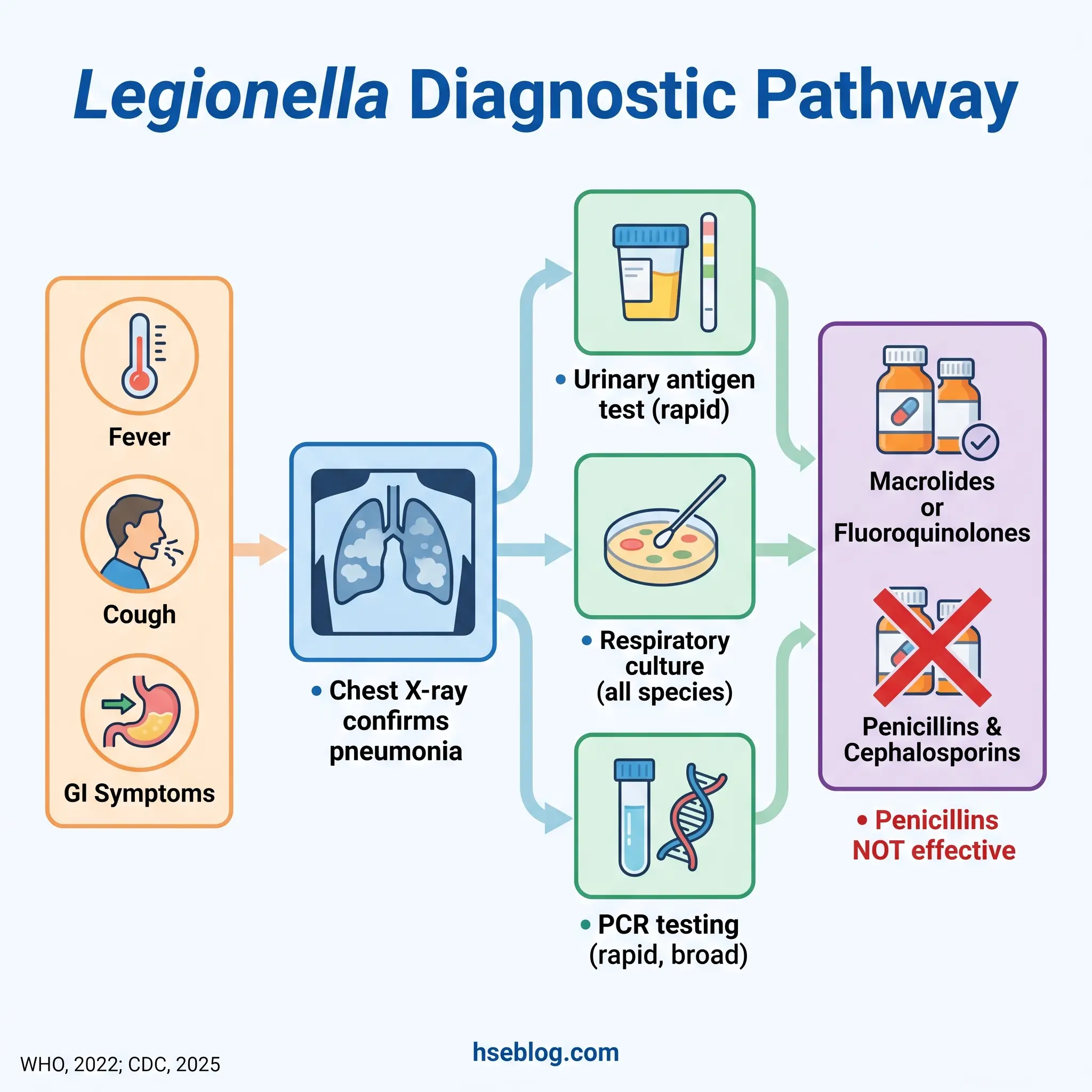
Diagnosis
A chest X-ray or CT scan confirms the presence of pneumonia but cannot identify the causative organism. Specific tests must be ordered — and this is where the diagnostic gap that drives chronic under-reporting sits.
The urinary antigen test is the most commonly used rapid test. It detects L. pneumophila serogroup 1 specifically, with sensitivity of 60–95% and specificity above 98% after day 3 of symptoms. Because serogroup 1 causes the majority of cases, this test catches most — but not all. Every non-serogroup-1 case and every non-pneumophila species will be missed by urinary antigen alone.
Culture of lower respiratory specimens (sputum, bronchoalveolar lavage) detects all Legionella species and serogroups. It takes 3–5 days to yield results but is essential for outbreak investigation because it allows molecular comparison between clinical and environmental isolates. PCR testing is increasingly used for rapid detection and can identify a broader range of species than urinary antigen.
The practical recommendation for both treating clinicians and public-health investigators: when Legionella is suspected, request both urinary antigen and lower-respiratory culture. Relying on urinary antigen alone contributes to the estimated 52,000–70,000 actual US cases versus the ~10,000 reported (National Academies of Sciences, Engineering, and Medicine, 2019).
Treatment
Effective antibiotics for Legionnaires’ disease are macrolides (azithromycin) and fluoroquinolones (levofloxacin). Penicillins and cephalosporins — the empirical first-line for many community-acquired pneumonias — are not effective against Legionella because the bacteria live inside host cells where these drug classes cannot reach. Early antibiotic initiation reduces mortality. Approximately one-third of hospitalised Legionnaires’ disease patients require intensive care.
How to Prevent Legionella: Core Control Strategies
Operational Legionella control in building water systems must be planned and supervised by a competent person with relevant training, jurisdiction-specific authorization, and site-specific risk assessment. The information here provides general HSE knowledge and does not replace that requirement.
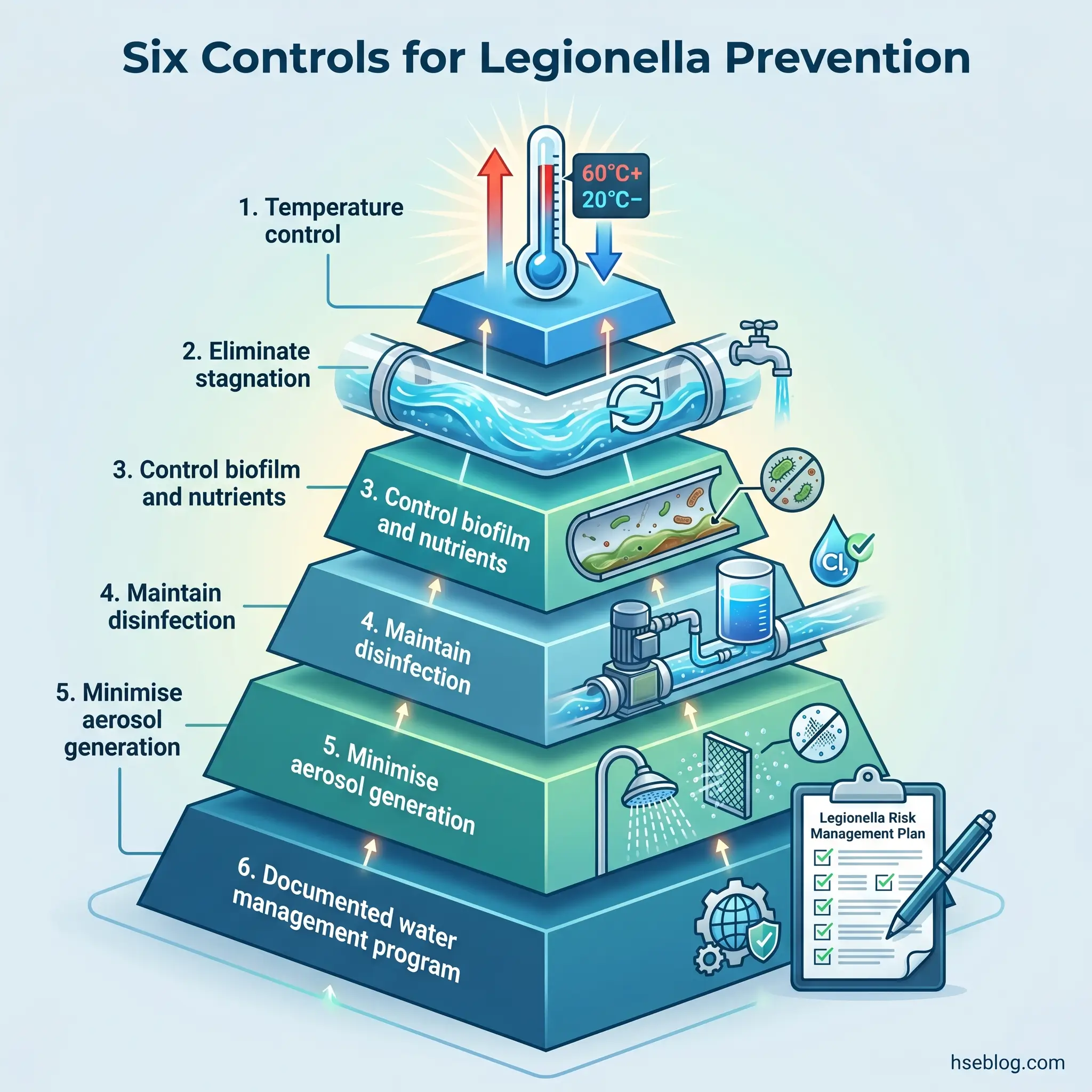
Legionella prevention in building water systems rests on a control hierarchy that targets each of the bacteria’s growth drivers. The controls below are well established across jurisdictions — what differs is the prescriptive detail and enforcement posture.
1. Temperature control — the single most important physical control. Hot water must be stored at temperatures that kill Legionella and delivered at temperatures that prevent growth. In the UK, HSE guidance (HSG274 Part 2) specifies hot water stored at ≥60°C and distributed so that it reaches ≥50°C within one minute at outlets. US CDC and ASHRAE guidance commonly cites ≥55°C delivery temperature. Cold water must be kept below 20°C (UK HSE guidance). Where a building operates across jurisdictions, the HSE-aligned 60°C storage / 50°C delivery / <20°C cold thresholds represent the stricter and safer default. Scald risk at these temperatures is real — thermostatic mixing valves (TMVs) are used to reduce outlet temperature to safe levels, but they require careful risk review because the mixed-water zone can itself sit in the Legionella growth range.
2. Eliminate stagnation — dead legs, infrequently-used outlets, and long pipe runs with low draw-off are where Legionella finds the still, warm conditions it needs. Weekly flushing of low-use outlets, removal or regular purging of dead legs, and careful management of buildings returning from closure are non-negotiable operational disciplines.
3. Control biofilm and nutrient sources — Legionella does not thrive in clean water alone; it lives within biofilm and feeds on other microorganisms. Material selection (WRAS-approved fittings in the UK), regular cleaning of storage tanks, descaling of shower heads and spray outlets, and sediment management in calorifiers all reduce the nutrient environment.
4. Maintain disinfection — chemical or physical. Cooling towers typically use biocides (chlorine, chlorine dioxide, bromine compounds) with residual monitoring. Building water systems may use chlorine dioxide, monochloramine, or copper-silver ionisation depending on the system and jurisdiction. UV treatment is effective point-of-use but does not maintain a residual in the distribution pipework. Thermal disinfection (pasteurisation at ≥70°C) is used for remediation.
5. Minimise aerosol generation — drift eliminators on cooling towers reduce the volume and particle size of water droplets released to atmosphere. Well-maintained, regularly-cleaned shower heads produce larger droplets that are less respirable.
6. Documented water management program — every control above must be part of a written plan that identifies hazards, assesses risk, defines control measures and their limits, assigns monitoring responsibilities, and establishes corrective-action procedures. This is the legal foundation under ACoP L8 (UK), the operational requirement under ANSI/ASHRAE Standard 188-2021 (US), and the risk-assessment-based approach embedded in EU Directive 2020/2184.
Legionella Risk Assessment: The Duty Holder’s Starting Point
Before any control measure is selected, a suitable and sufficient risk assessment must be completed. In the UK, ACoP L8 makes this an explicit legal duty for every duty holder whose premises contain a water system that could create a risk. In the US, ANSI/ASHRAE Standard 188-2021 begins with a building survey and risk assessment as the first step in any water management program. The 2021 edition of ASHRAE 188 rewrote previously permissive language as enforceable requirements, specifically to enable adoption into building codes and regulatory frameworks.
The risk assessment must be reviewed whenever the building use changes, the water system is modified, or an incident occurs — and at regular intervals regardless. A risk assessment that was completed at commissioning and never revisited is one of the most common findings in post-outbreak enforcement investigations.
Audit Point: The functional test of a risk assessment is not whether the document exists but whether it reflects the current state of the building, its population, and its water system. Ask the duty holder: “Has anything changed since this was last reviewed?” If the answer requires thought, the review is overdue.
Legionella Regulations: Legal Duties by Jurisdiction
No single global Legionella standard exists. Regulatory frameworks vary in structure, prescriptive detail, and enforcement mechanism, but converge on the same core principle: the person who controls a building’s water system bears legal responsibility for managing Legionella risk. The table below summarises the primary instruments across three major jurisdictions.
| United States | United Kingdom | EU/EEA | |
|---|---|---|---|
| Primary instrument | OSHA General Duty Clause (OSH Act §5(a)(1)); ANSI/ASHRAE 188-2021 (consensus standard) | Health and Safety at Work etc. Act 1974; COSHH 2002; HSE ACoP L8 (4th ed., 2013) | Directive (EU) 2020/2184 (recast Drinking Water Directive); national transposition laws |
| Key duties | Employers must protect workers from Legionella exposure; ASHRAE 188 requires building survey, risk assessment, WMP with control limits, verification and validation | Duty holders must identify/assess risk, prepare a written scheme, implement/monitor precautions, keep records, appoint a competent responsible person | Risk-assessment-based monitoring for Legionella in “priority premises”; parametric value <1,000 CFU/L |
| Healthcare-specific | CMS requires water management programs in all Medicare/Medicaid-certified healthcare facilities (since 2017) | ACoP L8 and HSG274 apply with heightened scrutiny for vulnerable populations | Member state implementation varies |
| Enforcement mechanism | OSHA citations; CMS Conditions of Participation; state/local laws (e.g., NYC Local Law 77 cooling-tower registration) | HSE prosecution; ACoP L8 has special legal status — failure to comply is admissible as evidence of breach | Member state enforcement; ECDC coordinates ELDSNet surveillance |
Several points deserve emphasis for duty holders managing across jurisdictions. First, the US lacks a single prescriptive federal standard — OSHA enforces through the General Duty Clause, and ANSI/ASHRAE Standard 188-2021 serves as the consensus benchmark. The 2021 revision specifically converted permissive language into enforceable requirements to support code adoption. Second, the UK’s ACoP L8 carries a legal weight that no US guidance document matches — in prosecution, a failure to follow the ACoP is treated as prima facie evidence that the duty holder did not do enough. Third, the EU’s Directive 2020/2184, effective from January 2023, introduced Legionella as a monitored parameter for the first time in European drinking-water law, with a parametric value of <1,000 CFU/L in priority premises.
The WHO’s foundational reference document — Legionella and the Prevention of Legionellosis (2007) — underpins all three frameworks. Its water-safety-plan methodology remains the international best-practice model.
Jurisdiction Note: Temperature thresholds are not universal. UK HSE specifies hot water stored ≥60°C and delivered ≥50°C with cold water <20°C. US CDC/ASHRAE guidance commonly cites ≥55°C delivery. The EU directive does not prescribe specific temperature targets but requires risk-assessment-based control. Duty holders operating across jurisdictions should default to the stricter UK thresholds where operationally feasible.
Why Legionnaires’ Disease Cases Are Rising
The global trajectory of reported Legionnaires’ disease cases is upward, and the acceleration is not solely attributable to better surveillance. In the US, reported cases grew roughly nine-fold between 2000 and 2018, peaking at 9,933 (CDC, 2025). The true disease burden is estimated at 52,000–70,000 annual US cases (National Academies of Sciences, Engineering, and Medicine, 2019). In the EU/EEA, the notification rate rose from 1.9 per 100,000 in 2020 to 2.6 in 2022 and reached 3.2 per 100,000 in 2023 — the highest ever recorded (ECDC’s most recent surveillance report, 2025).
The 2024–2025 period brought a cluster of high-profile outbreaks that reinforced the trend. A commentary in The Lancet Microbe (April 2025) formally flagged the 2024 global surge — spanning Melbourne, Lombardy, London, Albany (New York), New Zealand, and Hong Kong — as requiring heightened preparedness across public-health and building-management systems. The trend continued into 2025 and 2026: the Harlem (NYC) outbreak produced 114 cases and 7 deaths with twelve positive cooling towers (NYC Department of Health, 2025); Marshalltown, Iowa, London (Ontario), and Bloomingdale (Illinois) each reported significant clusters; and North Carolina documented a 54% year-on-year increase in cases, from 201 in 2024 to 310 in 2025 (NCDHHS, 2026).
The leading explanations among researchers and practitioners are not mutually exclusive. Ageing water infrastructure — particularly in cities with decades-old distribution networks and in-building plumbing — creates more opportunities for stagnation and biofilm. An ageing population concentrates more individuals in the high-risk demographic. Climate is a factor that has moved from theoretical to observable: warmer ambient temperatures extend the period during which cold-water systems drift above 20°C, expanding the Legionella growth window. Post-COVID building re-openings, where water sat stagnant for weeks or months, contributed a documented one-time spike that laid bare how many buildings lacked re-commissioning procedures. Improved diagnostic availability — wider use of urinary antigen testing — explains part of the increase but not the outbreak clustering.
The practical implication for facility and HSE managers is that Legionella control programs designed for 20th-century ambient conditions and occupancy patterns need reassessment against current realities. Buildings that coped historically with a design-ambient temperature of 20°C now see cold-water storage drifting above that threshold for longer stretches, and the engineering response — more insulation, active cooling of cold-water storage, revised monitoring frequencies — has not kept pace across the building stock.
Common Failures in Legionella Water Management
Published outbreak investigations and enforcement actions follow patterns that are remarkably consistent across jurisdictions, building types, and decades. The failures below are not hypothetical — they recur in HSE prosecution records, CSB-equivalent water-safety investigations, and public-health post-incident reports.
Risk assessments that exist in name only. The document was completed — often by a competent contractor — but then filed and never revisited. When the building changed use, added a wing, converted a floor, or brought in a new tenant population, no one triggered a review. The risk assessment described a building that no longer existed.
Temperature monitoring without trend analysis. Logs are completed and signed, but no one reads them for drift. A gradual 2°C rise in cold-water return temperature over six months — which can shift a system into the growth-favourable range — goes unnoticed until an outbreak investigation examines the records retrospectively.
Flushing regimes that collapse under real-world pressure. Weekly flushing of infrequently-used outlets is straightforward to specify and difficult to sustain. It is the first discipline to lapse during holiday periods, staff turnover, contractor handover, or when the person responsible moves to another role without a documented handover of water-safety duties.
TMV installations without post-installation risk review. Thermostatic mixing valves are installed to prevent scalding — a legitimate safety need. But the mixed-water zone downstream of a TMV can sit squarely in the 20–45°C growth range. A TMV retrofit without an updated Legionella risk assessment is a common finding in post-outbreak enforcement.
Cooling-tower biocide dosing failures. Automated dosing systems provide false assurance when the chemical drums run empty between service visits and no one checks. Manual dosing regimes depend entirely on the operator following the schedule, with no independent verification.
“Temporary” workarounds that become permanent. A disconnected outlet, a bypassed recirculation pump, a capped-off dead leg that was supposed to be removed during refurbishment — these remain in place for months or years, creating precisely the stagnation conditions Legionella requires.
The observable difference between a building that manages Legionella competently and one that does not is rarely visible in the paperwork. It becomes visible in the response when something goes wrong. Does a failed temperature reading trigger a documented corrective action within days, or does it disappear into a log that no one reviews? Duty holders should audit their own programme by asking what would happen — not what should happen.
Frequently Asked Questions

Conclusion
The regulatory and scientific consensus on Legionella control has never been stronger, yet the global case trajectory is moving in the wrong direction. The 2024–2025 outbreak surge — documented by The Lancet Microbe, tracked through ECDC surveillance showing the highest-ever EU/EEA notification rate, and visible in community clusters from Harlem to Melbourne — is not a statistical artefact of better testing. It reflects an ageing building stock, an ageing population, shifting climate patterns that push cold-water systems into growth-favourable temperatures, and a persistent gap between the existence of water management programs and their consistent, competent execution.
For duty holders and HSE professionals, the path forward requires treating Legionella risk as a living, continuously-managed operational discipline rather than a one-time compliance exercise. The standards — whether ANSI/ASHRAE 188-2021, ACoP L8, or EU Directive 2020/2184 — all demand the same fundamental cycle: identify, assess, control, monitor, record, review, and revise when conditions change. The buildings where that cycle operates with genuine rigour are not the ones appearing in outbreak reports.
The question worth asking of your own programme is not whether the documentation exists. It is whether, if a temperature reading failed tomorrow, a corrective action would be traceable within days — or whether it would disappear into a log that no one reads.

