In January 2011, a stainless-steel autoclave door failed on a 42-year-old UK engineer named Michael Whinfrey. The Health and Safety Executive investigation concluded that safety devices had been removed from the vessel to cut running costs — what the court described as systemic failings attributable to senior management. He had been standing in front of the door when it released under pressure. The head injuries were fatal.
The HSE had already been tracking a pattern. Its revised Guidance Note PM73 records two UK workplace fatalities since 2008 involving autoclaves — Whinfrey and an earlier incident in which a walkway collapsed during loading. Autoclaves are among the few pieces of routine laboratory equipment that combine three high-energy hazards in one vessel: pressurised steam, temperatures well above the boiling point, and biohazardous contents. Autoclave safety is therefore not a training topic you cover once and file. This guide covers the main autoclave hazards, the operating procedure that controls each one, the regulatory duties behind those procedures, and the monitoring that tells you whether a cycle actually worked.
What Is an Autoclave and Why Safety Matters
An autoclave is a pressure vessel that uses saturated steam at elevated temperature and pressure — typically 121 to 134 °C at 15 to 30 psi — to achieve sterilisation of loads. The same vessels run in hospital sterile processing departments, university research laboratories, pharmaceutical manufacturing, microbiology teaching benches, dental and tattoo studios, composite-curing shops, and biomedical waste treatment plants.
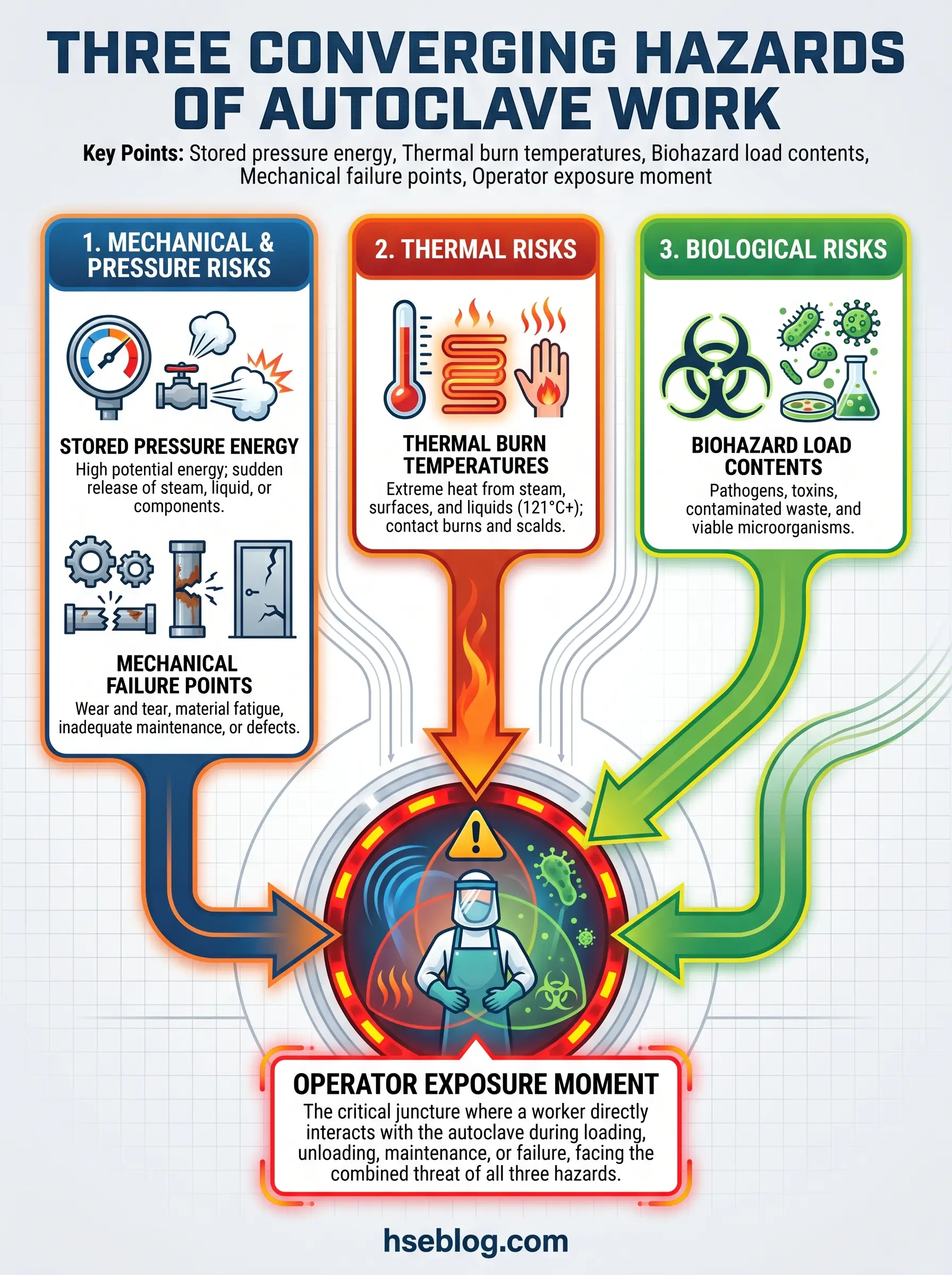
Three separate hazard physics run in parallel inside one stainless-steel chamber. Pressurised steam stores mechanical energy that, released violently, can propel a door or seal with projectile force. Saturated steam at working pressure sits at temperatures that inflict full-thickness burns on brief contact. And the load itself — cultures, contaminated sharps, infectious tissue — remains a biohazard until the cycle has demonstrably worked. That is why autoclave pressure vessel safety is not a niche subject for plant engineers; it is a daily operational duty for whoever cracks the door.
The Main Hazards of Autoclave Operation
The HSE classification of pressure-system hazards puts uncontrolled release of stored energy at the top — the violent ejection of components, pressurising medium, or vessel contents. Day to day, operators meet something duller and more frequent: heat. The autoclave hazards below are ordered by incident frequency across the sterilisation corridor I manage in a pharmaceutical QC facility, not by catastrophic potential.
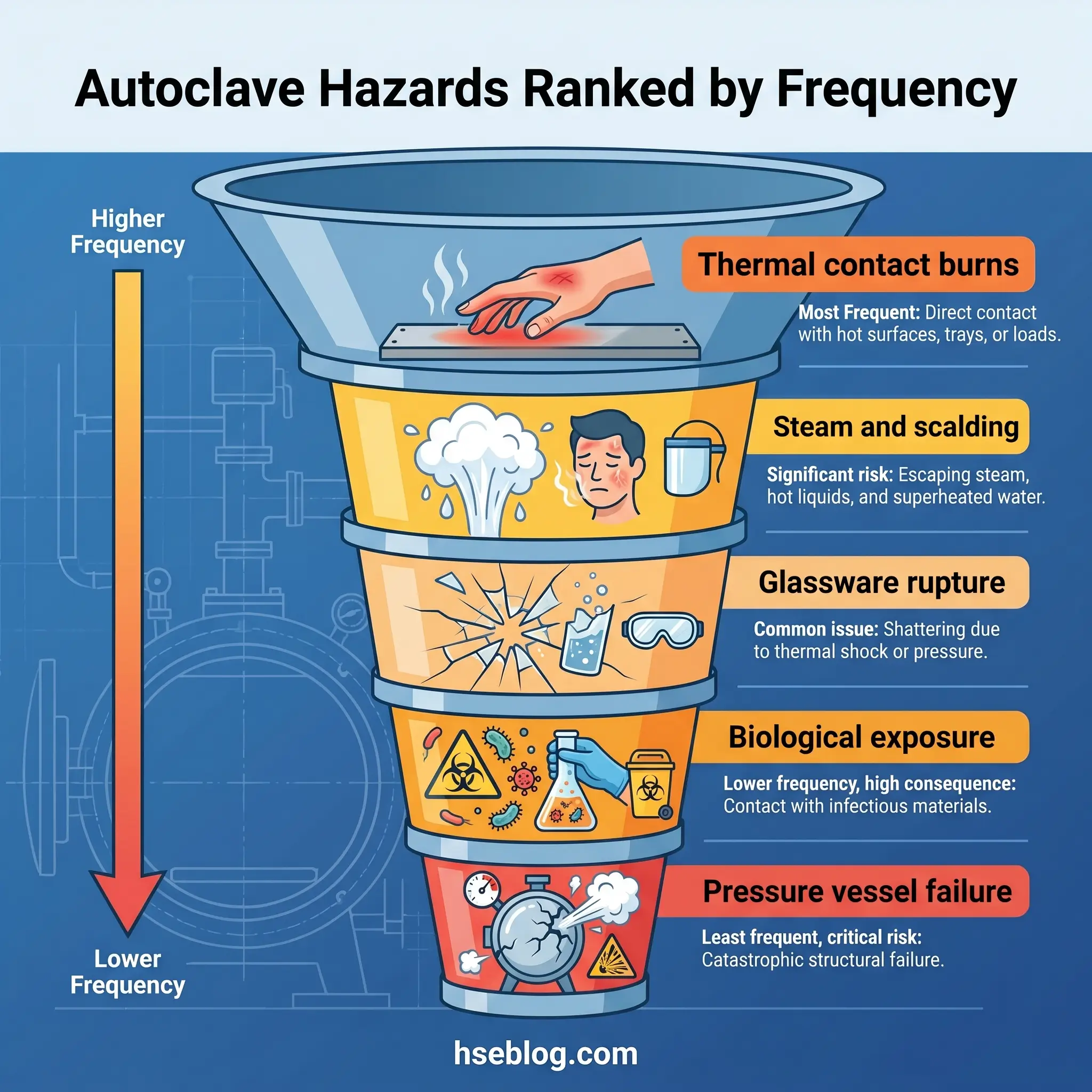
Thermal Burns from Hot Surfaces and Contents
The chamber wall, rack, door interior, and load sit well above 100 °C when the cycle ends, and they cool slowly. A gloveless hand brushing the inner door within five minutes of opening is the most common injury pattern on any working floor. The burns tend to be small, painful, and underreported — technicians run cold water and move on. They still matter, because they signal process drift: if operators are reaching in bare-handed, post-cycle cool-down time has been compressed by schedule pressure.
Steam Burns and Scalding
Steam escapes when the door is first cracked. It also escapes from sealed liquid containers that were not vented, and from autoclave bags whose necks loosened mid-cycle. A senior microbiologist I worked with described the residual steam release as “invisible until your forearm knows” — the plume is nearly clear, which is why the standard procedure has the operator stand to the side rather than in front. The Cornell phosphate-buffered saline rupture, where a hot bottle failed during removal, is taught widely because the operator was doing everything else right; the bottle was simply jarred as she set it on the bench.
Pressure Vessel Failure and Violent Door Opening
This is the low-frequency, high-consequence failure. Door interlocks are the primary engineered control, and HSE PM73 is explicit on two requirements: the door must be positively secured before pressurisation can begin, and nil pressure inside the vessel must be verified before the door can be released. The Whinfrey fatality sits in the background of every serious conversation about interlocks — safety devices were removed, and a door blew. Operators who bypass interlocks for convenience, or managers who allow bypassed interlocks on a production floor, are replicating the same chain of decisions.
Exploding Glassware and Container Rupture
Flasks, bottles, and Schott containers fail when caps are sealed, when the glass is cracked, or when rapid cooling produces thermal shock. Sealed caps are the dominant mechanism: trapped pressure equalises with the chamber during the cycle and then exceeds the glass’s strength during depressurisation. The two-thirds fill rule and a loosened or vented closure are not optional — they are the difference between a successful cycle and a hot-bottle explosion.
Fire and Chemical Reactions
Oxidiser plus organic plus heat is a known explosion pathway in pressure vessels. The historical precedent operators cite is a hydrogen-oil autoclave-cell fireball, but the everyday mistake is simpler: bleach-contaminated waste goes into a biohazard bag and autoclaves alongside cultures. The result ranges from a chemical spatter to a chamber fire. Autoclaves are sterilisation devices, not chemical disposal units — chemically contaminated waste belongs in its own disposal stream.
Biological Exposure from Incomplete Inactivation
A cycle can fail mechanically, logically, or biologically. The load may not reach temperature, air pockets may prevent steam penetration, or the organism may be resistant. Prions are not inactivated by standard steam cycles. Certain biotoxins survive. If the cycle fails and the load is handled as sterile, exposure occurs at the glove-off step, hours later, with no warning.
Ergonomic and Slip/Fall Hazards
Heavy trays, overhead reaching on stacked sterilisers, and wet floors from condensate produce the injuries nobody writes about. The HSE walkway-collapse fatality involving autoclave loading is a reminder that the approach to the chamber — platform, stair, floor surface — is part of the safe system of work, not scenery.
Who Can Use an Autoclave: Training and Competency Requirements
The practical question new supervisors ask is not whether training is required but what has to be inside it. The outer frame is set by PUWER regulation 9 in the UK and 29 CFR 1910.132 in the US, but those texts give duties, not curricula. The curriculum has to come from the facility.
Training on any specific autoclave needs to cover manufacturer-specific operation for that model (a benchtop Tuttnauer and a 400-litre double-door Getinge are not interchangeable), cycle-to-load matching, loading limits and stacking rules, hazard recognition drawn from the hazard list above, emergency stop location and function, spill response procedures, and PPE donning and doffing for both solid and liquid loads.
Training is documented — signed by the trainer and the operator, held by the supervisor, reviewed at onboarding and after any incident, equipment change, or SOP revision. Competency verification is observational: the operator runs a full cycle, including loading and unloading, under the trainer’s eye, and the trainer signs off when the performance is correct without prompting. I do not release a new operator to solo use until I have watched them handle an unloading of hot liquids. That is the step where mistakes become injuries.
Required Personal Protective Equipment (PPE)
Standard lab PPE is the floor, not the ceiling. The PPE for a run of autoclave tips and dry glassware is not the PPE for a run of ten-litre media bottles. The table below maps each item of autoclave PPE to the hazard it is chosen to control and flags when it is required beyond minimum.
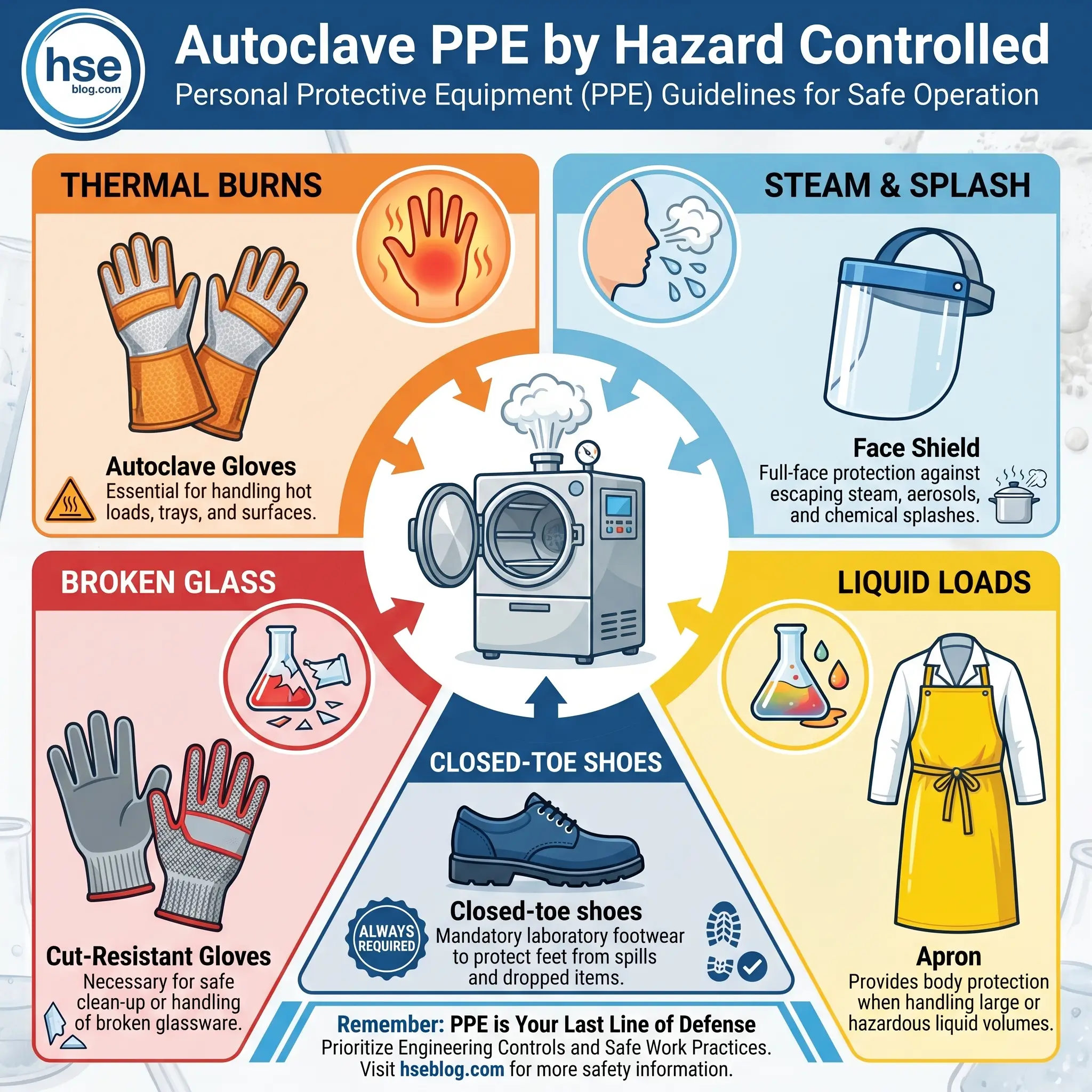
| PPE item | Hazard controlled | Required when |
|---|---|---|
| Buttoned lab coat, closed-toe shoes, safety glasses | Splash, broken glass underfoot, chemical carryover | Every cycle, every load |
| Heat-resistant autoclave gloves past the wrist | Thermal burn from chamber surfaces and loads | Every loading and unloading of a warm chamber |
| Full-length face shield | Steam burn to face, splash from liquid rupture | Any liquid load; any decontamination bag with visible liquid |
| Rubber or splash apron | Scalding from container failure | Any liquid load above about 500 mL total |
| Cut-resistant gloves or tongs | Lacerations from broken glass | Post-cycle glass breakage, sharps handling |
| Face mask | Airborne particulate in decontamination room | Per AAMI ST79 clause 4.5.2 in decontam environments |
Gloves lose insulation over time. I replace autoclave gloves when the palm material feels thin or the cuff has scorched, not on a calendar schedule. The OSHA QuickFacts on laboratory autoclaves and sterilisers emphasises the employer’s duty under 29 CFR 1910.132 to assess thermal and laceration hazards and provide PPE that matches — a line worth keeping visible in any PPE SOP.
Pre-Operation Checks and Loading Procedures
The loading procedure is the first half of an autoclave standard operating procedure and the point at which a drifting cycle becomes a failed cycle. Each step below ties to a hazard the earlier sections defined.
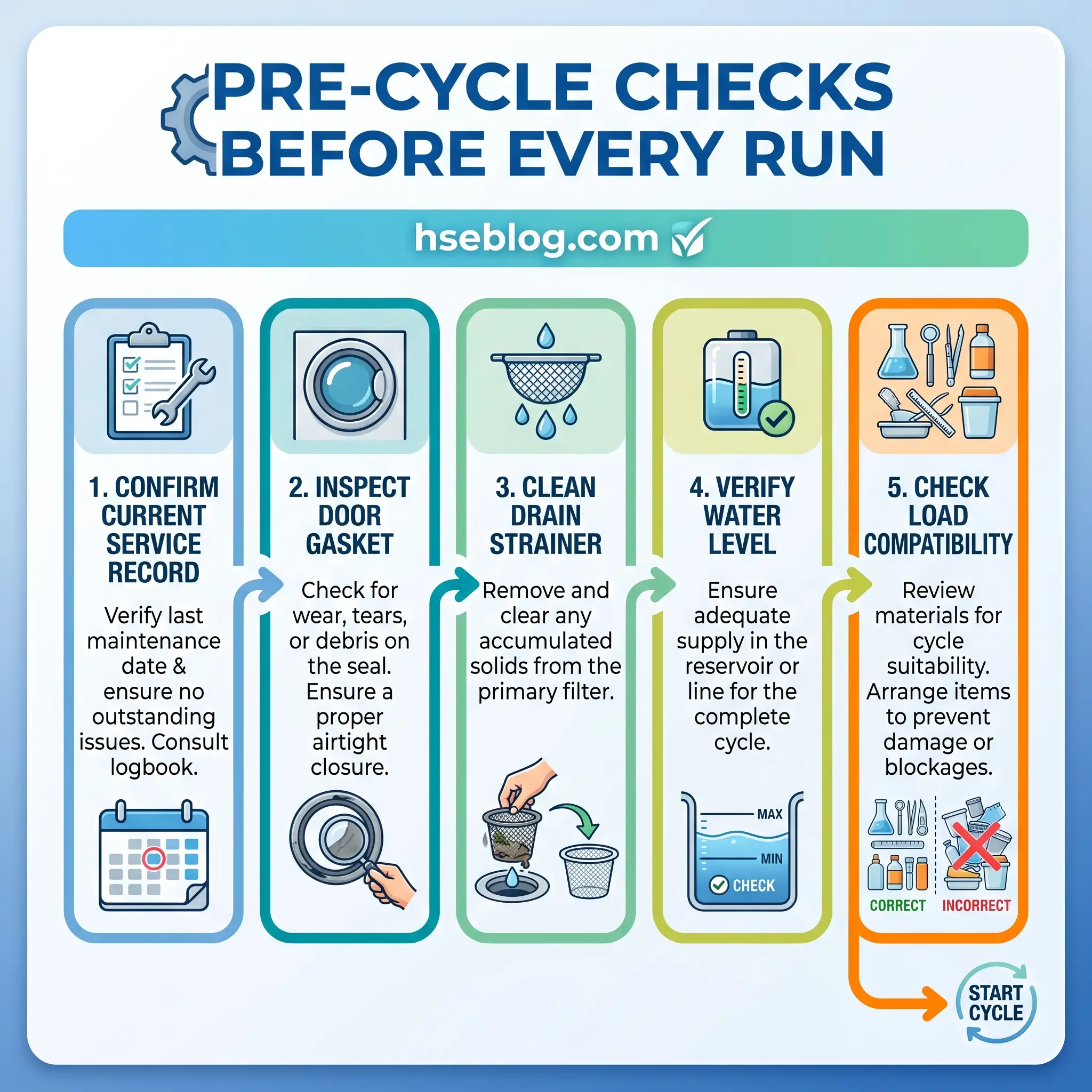
- Confirm the unit has a current service and examination record and is not carrying an “out of service” tag.
- Open the door and check the chamber for items left by the previous user — broken glass, residue, forgotten trays.
- Inspect the door gasket for cracks, bulges, thinning, or debris embedded in the seal face. A compromised gasket is the most common reason a cycle loses pressure mid-run.
- Clean the drain strainer. A blocked strainer traps condensate, shifts the internal pressure profile, and can foul steam purge on a gravity displacement cycle.
- Confirm the water reservoir level on a benchtop unit, or jacket pressure on a large unit, reads within the manufacturer range.
- Verify chamber and jacket pressure gauges at expected standby values before loading.
- Confirm the load is compatible: stainless 304/316, Pyrex, polypropylene, and polycarbonate are autoclavable. PVC, polystyrene, HDPE and LDPE, nylon, paper without secondary bagging, sealed containers, flammables, oxidisers, hazardous chemicals, radioactive material, and certain biotoxins and prions are not.
- Load into a stainless-steel or autoclavable plastic secondary containment tray. A spill inside the chamber becomes a biohazard clean-up, not a wipe-down.
- Leave space between items for steam circulation, position biohazard bags on their side to aid steam penetration, and do not let bags touch chamber walls.
- Fill liquid containers to no more than two-thirds, loosen all caps, or use vented closures. No sealed container enters a chamber that is about to pressurise.
- Apply autoclave indicator tape across the biohazard symbol on every bag so a glance at the waste post-cycle tells you whether it actually ran.
What You Should Never Put in an Autoclave
Operators continue to autoclave materials that damage chambers, poison colleagues, or ignite. The list below is not a puritanical ban — it is the shortest version of “what actually fails under steam at pressure,” and it needs to be visible in the facility SOP rather than buried inside loading rules.
- Flammable liquids and solvents — vapour ignition and explosion risk
- Oxidisers including bleach — oxidiser plus organic plus heat is a documented explosion pathway
- Corrosives and volatile acids — vapour damages chamber, gaskets, and operator
- Radioactive material — belongs in the radiation protocol, not the steam cycle
- Sealed containers of any kind — pressure rupture at depressurisation
- Sharps outside puncture-resistant containers — post-cycle glove penetration
- Non-autoclavable plastics — PVC, polystyrene, standard PE, nylon, acrylic
- Animal carcasses and chemically contaminated waste — dedicated disposal streams
- Powders, oils, and waxes — moist heat does not penetrate; choose dry heat or an alternative process
Watch For: Bleach poured into a biohazard bag “to disinfect before autoclaving” is the single most common oxidiser error I see on audit. The operator believes they are doubling protection. They are creating the reaction.
Running the Cycle: Choosing Parameters and Monitoring
Cycle selection is where the load pattern meets the physics. A wrapped surgical pack and a flask of culture broth behave differently in the same chamber, and running both on one cycle will sterilise neither well. Three cycle types cover routine laboratory and clinical use.
| Cycle type | Air removal | Typical parameters | Suited to |
|---|---|---|---|
| Gravity displacement | Passive — steam displaces air downward | 121 °C, 15 psi, 15–30 min (60 min for biohazardous waste) | Open containers, glassware, dry solids, non-porous loads |
| Pre-vacuum | Pulsed vacuum pumps remove air before steam | 132–134 °C, 27–30 psi, 3–10 min | Wrapped instruments, porous and textile loads, lumened items |
| Liquid cycle | Gravity removal with slow exhaust ramp | 121 °C, 15 psi, 20–60 min by volume | Liquids of any kind — never a vacuum-dry cycle for liquids |
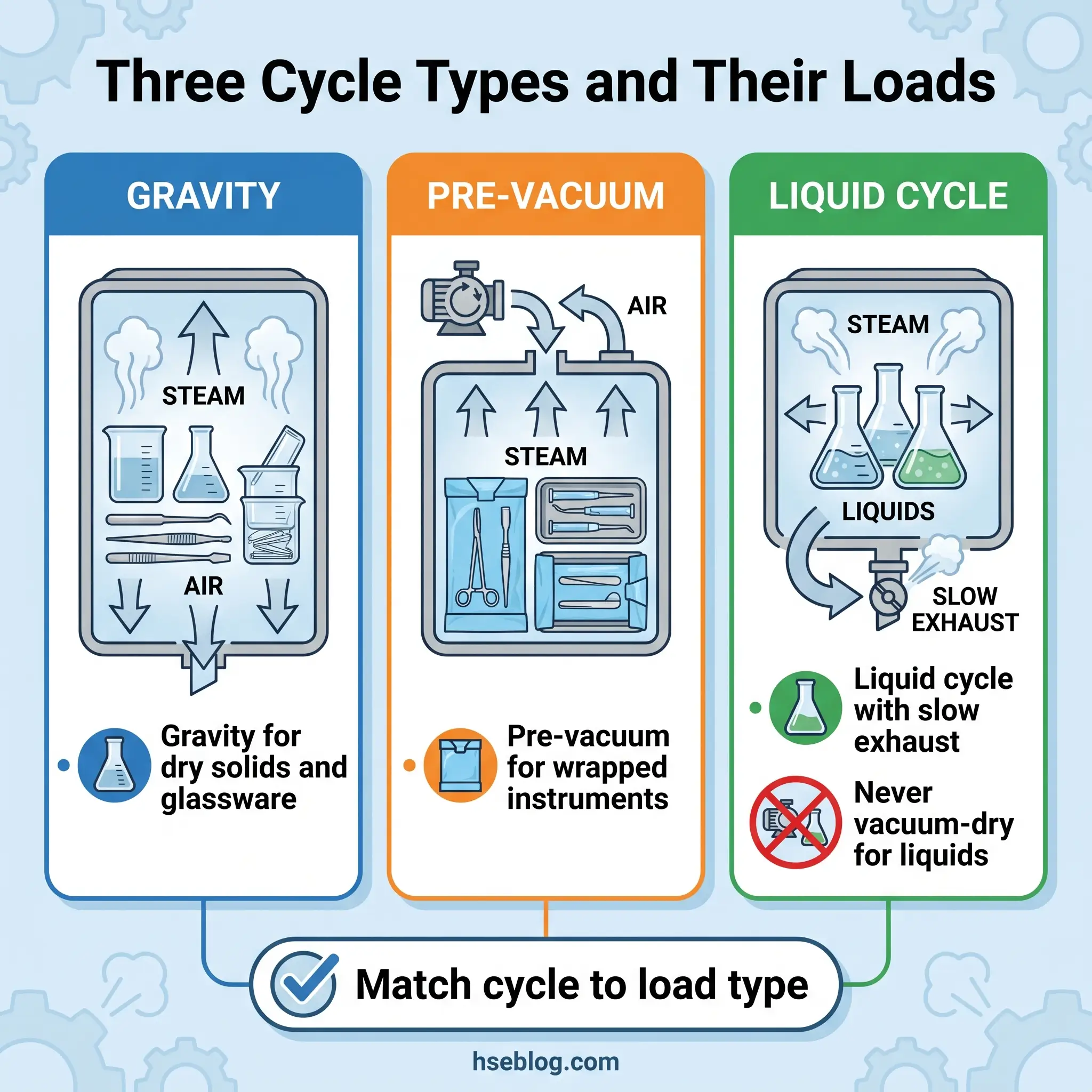
Monitoring runs as a hierarchy, and mixing the tiers is one of the ways sterile processing departments drift into sterility assurance trouble. Mechanical monitoring — the printed cycle record and gauge readings — tells you what the chamber did. A chemical indicator (autoclave tape) confirms the load reached steam temperature. A chemical integrator gives a time-at-temperature confirmation. A biological indicator using Geobacillus stearothermophilus spores tells you the cycle killed the toughest reasonable challenge organism. Only the last of those four is a sterility assurance claim.
The spores chosen for biological indicators are not incidental. Geobacillus stearothermophilus survives 121 °C for five minutes and is killed within thirteen — that resistance margin is why it is the reference organism across ISO 11138 and AAMI ST79. CDC guidance for healthcare facilities sets the floor at weekly biological indicator monitoring, with every load containing an implantable device, and the monthly minimum for biohazardous waste cycles set by state EPA rules.
A positive biological indicator is not an inconvenience — it is a recall event. The CDC Table 12 protocol for managing a positive biological indicator requires removal from service, load recall, and three consecutive negative BIs before return to service. It is worth reading that document cold and writing your own version before you need it, because “we will figure it out when it happens” is not a plan your quality auditor will accept.
Safe Unloading: The Highest-Risk Moment
More autoclave injuries happen at unloading than at any other point in the cycle, which is why treating unloading as an extension of loading — with the same casualness — is a documented route to the emergency department. This is the step where standing in the wrong place, rushing the cool-down, or trusting apparent stillness produces burns.
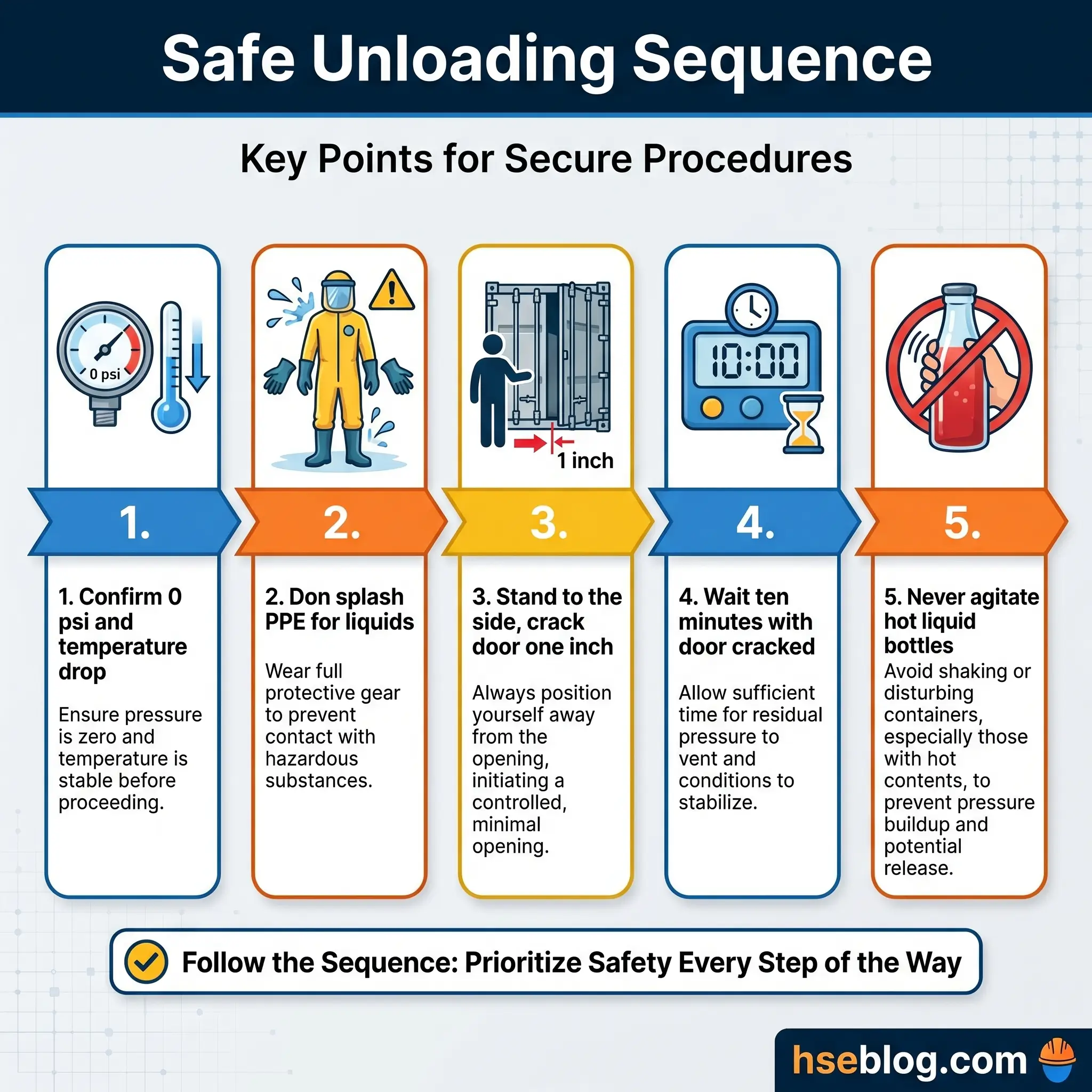
- Confirm the chamber reads 0 psi and the temperature has dropped to 121 °C or below before the interlock releases. On a modern unit the door will not unlock earlier; on an older unit with a manual override, do not override it.
- Don splash PPE if the load contained liquids — face shield and apron over the standard set.
- Stand to the side of the door, not in front, and crack the door no more than an inch (about 2.5 cm) for ten seconds to vent residual steam.
- Wait ten minutes with the door cracked for trapped air to escape and contents to equilibrate. This is the window that prevents hot-bottle explosion.
- Wait a full hour before any ungloved contact with liquid containers. Superheated liquids — those above their atmospheric boiling point — hide behind apparent stillness and flash-boil when disturbed.
- Never agitate, transport, or uncap hot liquid containers. Transport by trolley, cap off, at a walking pace.
- Place hot items in a designated hot zone with signage. Nobody should be able to brush past a cooling tray without warning.
- Clean condensate from the floor immediately. The slip hazard is the one that catches observers, not operators.
- Log the cycle: date, operator, contents, cycle type, indicator result, anomalies. The logbook is the operator’s audit defence.
Autoclave Maintenance and Examination Schedule
Regulation 8 of the Pressure Systems Safety Regulations 2000 splits autoclave maintenance into two legally distinct categories that SOP writers routinely conflate. User-level tasks — the daily-to-monthly work the operator performs — keep the unit running. A competent-person thorough examination under a Written Scheme of Examination — independent, qualified, and scheduled — keeps the employer out of a prosecution bundle.
| Interval | Task | Performed by |
|---|---|---|
| Daily | Wipe interior, visual gasket check, record cycle in logbook | Operator |
| Weekly | Clean drain strainer, empty-chamber test cycle, visual gasket inspection | Operator |
| Monthly | Detailed gasket, door, shelf and wall inspection; BI validation where required | Operator with supervisor |
| Semi-annual | Temperature probe calibration against NIST-traceable reference | Qualified technician |
| Annual | Full service visit, safety valve functional test, interlock verification | Manufacturer or authorised service engineer |
| 12–24 months | Scheduled door gasket replacement | Manufacturer or authorised service engineer |
| Per WSE | Thorough examination by competent person | Independent competent person (PSSR reg 8) |
Temperature probe calibration at six-month intervals is the GMP norm under 21 CFR 211.68 and EU GMP Annex 15; outside GMP environments, annual calibration remains defensible for research use. ASME Boiler and Pressure Vessel Code Section VIII Division 1 requires the pressure relief valve to operate within ±2 percent of its set pressure, which is why the annual valve functional test is not a checkbox — it is a verification against code.
In the UK, the Written Scheme of Examination must be drawn up by a competent person before a qualifying pressure system is put into use. The WSE specifies which parts are examined, the nature of each examination, and the maximum interval between examinations. For autoclaves above the 250 bar-litre threshold, running without a current WSE is an enforcement matter, and the HSE PSSR landing page remains the primary reference for duty-holders writing or renewing one. Approved Code of Practice L122 gives HSE-approved guidance on the independence and authority of the competent person, including the power to stop the equipment being used when unsafe.
Emergency and Incident Response
Most competitor guides stop at prevention. The field reality is that cool heads in the first sixty seconds of an incident change the outcome. The responses below are triage-level and sit in front of the full investigation process, not instead of it.
Burn or scald injury. Cool running water for ten to twenty minutes. Do not apply ice. Any burn larger than the casualty’s palm, any burn with blistering, and any facial, respiratory, or third-degree burn is a medical emergency — call for transport.
Chamber spill. Wait for full cool-down before entering the chamber. Don PPE matched to the residual hazard. Use an absorbent material or spill kit; place broken glass and sharps in a puncture-resistant container. Decontaminate the spill area per the site biological spill procedure. Document.
Failed cycle or positive biological indicator. Tag the unit “Out of Service.” Do not release the load. Notify the supervisor and initiate the CDC Table 12 and AAMI ST79 positive-BI protocol. Three consecutive negative BIs return the unit to service.
Visible equipment damage — door warping, steam from a closed door, water pooling under the unit, unexplained pressure loss. Isolate electrical and steam supply. Tag out. Request qualified service. Do not attempt to nurse it through one more cycle.
Report all incidents and near-misses to the supervisor and EHS per local policy. The near-miss that never reaches the log is the near-miss that becomes next year’s incident.
Regulatory Framework and Standards
Autoclave operation sits at the intersection of pressure-system law, PPE law, and healthcare sterilisation standards. The specific citations matter because SOPs that reference them are defensible in audit and in prosecution.
In the United States, OSHA’s General Duty Clause 5(a)(1) establishes the baseline duty. 29 CFR 1910.132 requires hazard assessment and appropriate PPE — the framework OSHA QuickFacts on laboratory autoclaves applies specifically to thermal and laceration exposures. Under 29 CFR 1910.1030 paragraph (e) — the Bloodborne Pathogens Standard — failure to provide an autoclave for decontamination of regulated waste within or as near as possible to the work area in HIV and HBV research laboratories is a citable condition.
In the United Kingdom, the Pressure Systems Safety Regulations 2000 are the central duty framework, with Regulation 8 establishing the Written Scheme of Examination and Approved Code of Practice L122 providing HSE-approved guidance. The Provision and Use of Work Equipment Regulations 1998 (PUWER) sits alongside PSSR for operational use. HSE Guidance Note PM73 is the specific document on autoclave safety — covering door interlocking, nil-pressure verification before opening, fail-safe automated controls on loss of power or over-pressure or over-temperature, and safeguards against inadvertent pressurisation with a person inside a walk-in autoclave.
Pressure vessel construction is governed by ASME Boiler and Pressure Vessel Code Section VIII Division 1 in North America, with the “U” stamp marking a compliant vessel and the National Board “R” stamp marking authorised alterations. In the EU, Directive 2014/68/EU on pressure equipment applies, with conformity assessment and CE marking, and EN 13445 serving as the harmonised standard for unfired pressure vessels.
For sterilisation itself, ANSI/AAMI ST79 is the comprehensive guide to steam sterilisation and sterility assurance in healthcare facilities. The current version is the 2017 edition reaffirmed in 2022, and AAMI opened the full revision process in September 2025 — the first comprehensive update in nearly a decade, addressing technology changes, harmonisation with ST91 on flexible endoscope processing, and updated monitoring. Any sterile processing lead should track that consultation rather than be surprised by the published version. AAMI also published TIR109:2025 on external transport of reusable medical devices for processing in March 2025 — relevant to hub-and-spoke sterilisation models where a central facility autoclaves for satellite clinics.
ISO 17665 sets the international requirements for the development, validation, and routine control of the moist-heat sterilisation process, targeting the SAL of 10⁻⁶ that defines sterile. ISO 11138 governs biological indicators and ISO 11140 governs chemical indicators — both are referenced inside AAMI ST79 and are the testable standards behind the tape, strips, and vials on the chamber shelf.
Audit Point: Auditors do not find missing standards by looking for missing standards. They find them by reading the facility SOP and asking “which regulation says you have to do it that way?” Write the citation into the SOP at the step it justifies, and that question becomes a one-breath answer.

Frequently Asked Questions
Conclusion
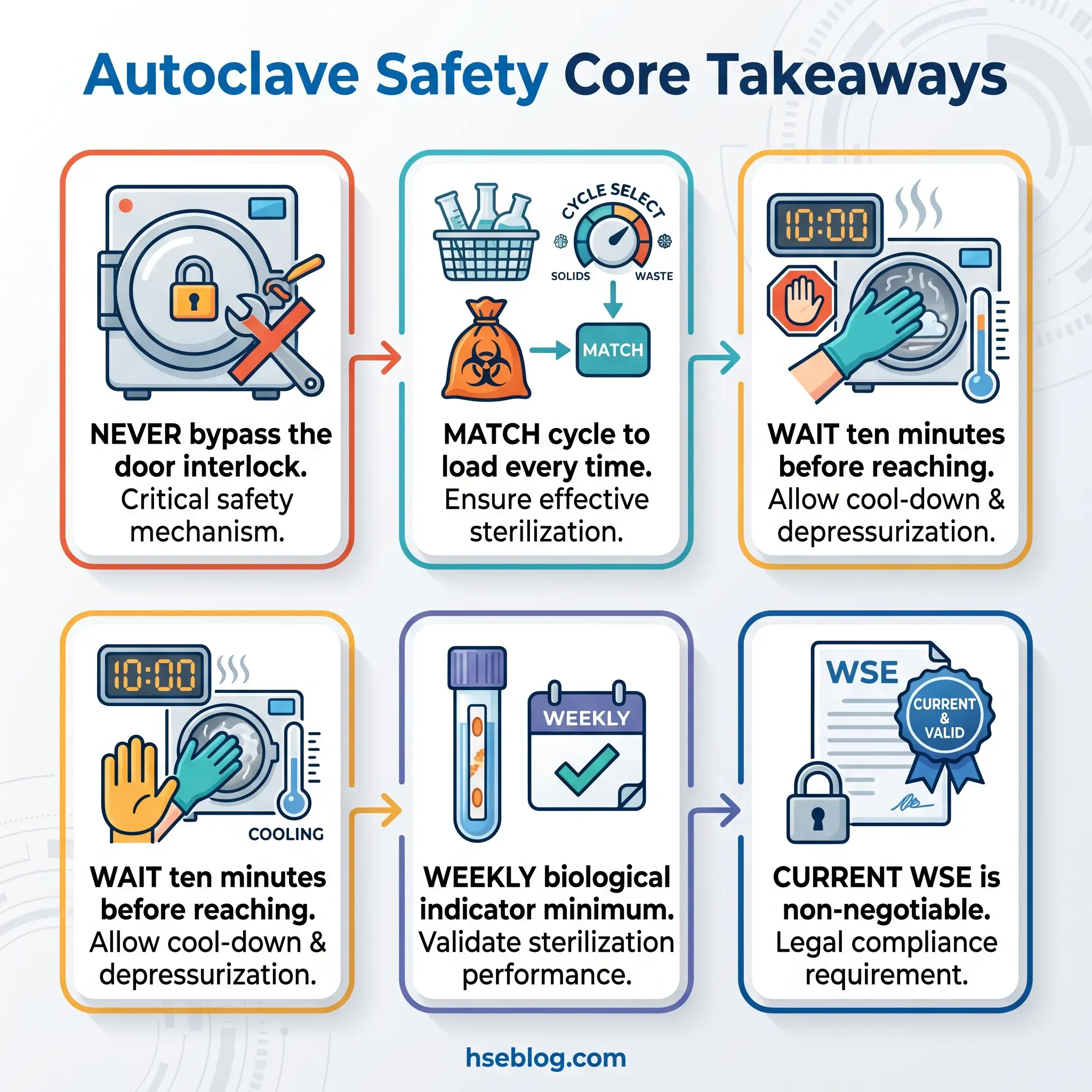
The industry’s consistent mistake with autoclaves is to treat them as sterilisers first and pressure vessels second. The order is backwards. Every operational control — the interlock, the nil-pressure verification, the Written Scheme of Examination, the relief valve test — exists because the vessel stores enough energy to kill the person standing in front of it. Sterility assurance is the purpose; pressure vessel safety is the floor you build it on.
The single highest-impact change a facility can make is not a new autoclave or a new indicator — it is making the door interlock and the Written Scheme of Examination visibly non-negotiable. That means walking the sterilisation corridor and asking whether any operator on any shift has learned that a safety override can be pressed without consequence, whether the WSE binder carries a current signature, and whether the last positive biological indicator triggered a full Table 12 response or a quiet re-run. If those three answers are clean, the catastrophic tail risk is controlled. If any of them is soft, the conditions behind the Whinfrey fatality are in the building.
Autoclave safety is not earned once. It is earned every shift, at the door, by the person who waits the ten minutes.

