TL;DR
- If air monitoring is your only exposure assessment tool → you are likely underestimating true dose for any chemical with a skin notation, because dermal and ingestion routes bypass the sampling pump entirely.
- If your workers handle lead, cadmium, or benzene → check your jurisdictional mandate — OSHA requires biological monitoring for these three substances, but Cal/OSHA’s 2025 revised lead standard now triggers action at dramatically lower blood lead levels than the federal rule.
- If biological monitoring results exceed a guidance value → this is a control-system failure signal, not a medical diagnosis — initiate a review of engineering controls, work practices, and PPE before the next sampling round.
- If you treat biological monitoring as your entire health surveillance programme → you have a gap — biological monitoring measures dose, not health effect, and it does not replace clinical assessment or biological effect monitoring.
Biological monitoring is the measurement of a chemical substance, its metabolites, or its biological effects in a worker’s body fluids — typically blood, urine, or exhaled breath — to assess how much of a workplace chemical has actually been absorbed. It is needed when air monitoring alone cannot reliably capture total exposure, particularly for chemicals absorbed through the skin, when PPE effectiveness requires verification, or when multiple exposure routes operate simultaneously.
Federal OSHA mandates biological monitoring for exactly three chemical substances — lead, cadmium, and benzene after an unplanned release (NIOSH, 2022). That number surprises most safety professionals who assume the regulatory net is far wider. The gap between those three mandated chemicals and the roughly 30 agents for which ACGIH publishes voluntary Biological Exposure Indices reveals something uncomfortable: for the vast majority of workplace chemicals where biological monitoring would add genuine protective value, it remains a professional-practice decision rather than a legal requirement — and that decision goes unmade far too often.
The consequence is measurable. Workers handling isocyanates, organophosphates, chromium (VI), or mercury may have their airborne exposure carefully sampled while the dominant absorption pathway — through unbroken skin, contaminated food, or incidental hand-to-mouth contact — goes entirely unassessed. Biological monitoring closes that gap by measuring what has actually entered the body, regardless of route. This article covers what biological monitoring is, how it differs from air monitoring and health surveillance, the biomarkers and matrices used, the specific regulatory triggers across US, UK, and EU jurisdictions, how to build an effective programme, and the ethical framework that governs the collection of biological samples from workers.
What Is Biological Monitoring?
Biological monitoring is the systematic measurement of a chemical agent, its metabolites, or the product of an interaction between the agent and a worker’s body, in biological specimens collected from exposed individuals. The specimens are typically blood, urine, or exhaled breath. The purpose is to assess internal dose — how much of a substance has actually been absorbed — rather than external concentration in the breathing zone.
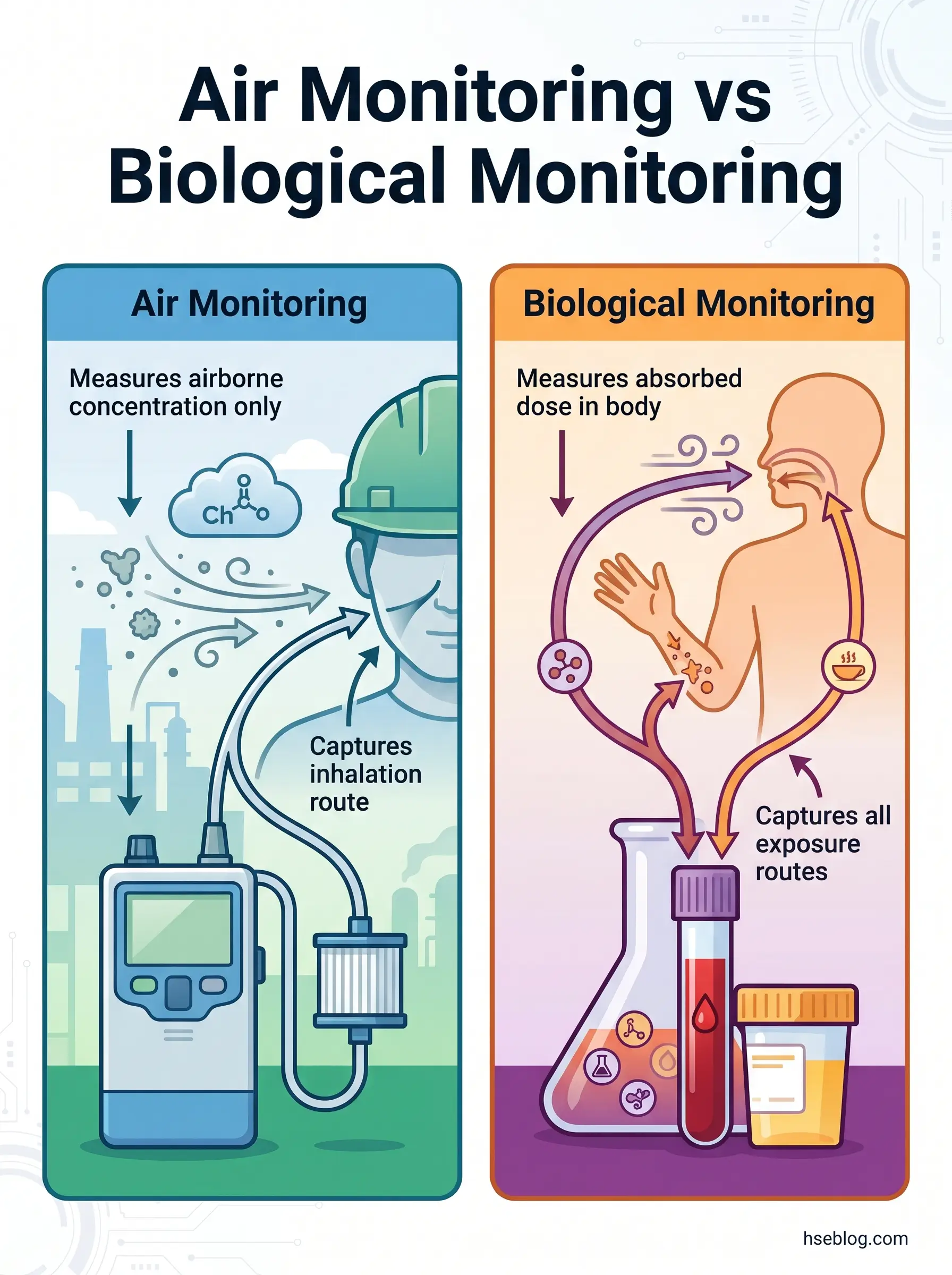
This distinction is fundamental. Air monitoring, the more familiar exposure assessment tool, measures the concentration of a contaminant in the worker’s immediate environment. It captures what is available to be inhaled. Biological monitoring captures what has actually crossed into the body, through every route — inhalation, skin absorption, and ingestion combined. For chemicals that carry a skin notation in occupational exposure limit tables, this difference is not academic. A worker handling toluene diisocyanate (TDI) with adequate respiratory protection but inadequate glove selection may return personal air monitoring results well within the occupational exposure limit while absorbing a significant dose through the skin. Only biological monitoring — in this case, measuring urinary diamine metabolites — would reveal that the control regime has failed.
The practical value extends beyond skin-absorbed chemicals. Biological monitoring also accounts for individual variability in absorption, metabolism, and work practices that air monitoring cannot capture: differences in breathing rate, the actual seal quality of a respirator across a shift, eating or smoking in transition zones, and the cumulative effect of overtime hours that extend beyond the sampling period.
How Biological Monitoring Differs from Air Monitoring
Air monitoring and biological monitoring answer different questions. Air monitoring answers: what concentration exists in the work environment? Biological monitoring answers: how much of this substance has the worker’s body actually absorbed?
The practical implications of that difference shape when each tool is appropriate. Air monitoring is the right starting point for assessing workplace conditions, verifying ventilation performance, and comparing environmental concentrations against occupational exposure limits. It is essential, and nothing here diminishes its role. But it has inherent blind spots.
Air monitoring cannot account for dermal absorption — and for chemicals carrying a skin notation in EH40, the ACGIH TLV booklet, or equivalent national OEL lists, dermal uptake may equal or exceed the inhalation dose. It cannot account for ingestion from contaminated hands, food, or cigarettes. It cannot tell you whether the respirator a worker wore for eight hours actually sealed properly for all eight hours. And it cannot capture the individual biological variability that determines how much of an inhaled dose crosses the alveolar membrane into blood.
Biological monitoring fills those gaps. When both tools are used together, the exposure assessment gains a completeness that neither provides alone. NIOSH guidance on applying biological monitoring methods reinforces this complementary relationship — biological monitoring is not a replacement for air monitoring but a parallel tool that validates whether controls are protecting the worker, not just the work environment.
How Biological Monitoring Relates to Health Surveillance
Three terms create persistent confusion in practice: biological monitoring, biological effect monitoring, and health surveillance. They are related but not interchangeable, and treating them as synonyms leads to programme gaps that auditors — and more importantly, actual health outcomes — will eventually expose.
Biological monitoring measures the dose of a chemical in the body. It tells you how much substance was absorbed. A blood lead level is biological monitoring. Urinary mercury concentration is biological monitoring. The question it answers is: did the worker absorb this chemical, and at what level?
Biological effect monitoring measures early, reversible biological changes caused by chemical absorption. It tells you what the substance is doing to the body. Zinc protoporphyrin levels indicating lead’s interference with haemoglobin synthesis — that is biological effect monitoring. Cholinesterase depression from organophosphate exposure — also biological effect monitoring. The question it answers is: is this substance causing a measurable effect?
Health surveillance is the broader programme that encompasses both of the above, plus clinical examination, health questionnaires, and fitness-for-task assessment. HSE UK guidance on biological monitoring and health surveillance draws this distinction clearly — biological monitoring is a tool within health surveillance, not a substitute for it.
A failure pattern that recurs across published programme reviews: organisations collect urine or blood samples on the correct schedule, file the results, and consider their health surveillance obligation met. No clinical assessment. No trending of results over time. No feedback loop into the risk assessment. The biological monitoring data exists, but the programme architecture around it is absent.
Types of Biomarkers Used in Biological Monitoring
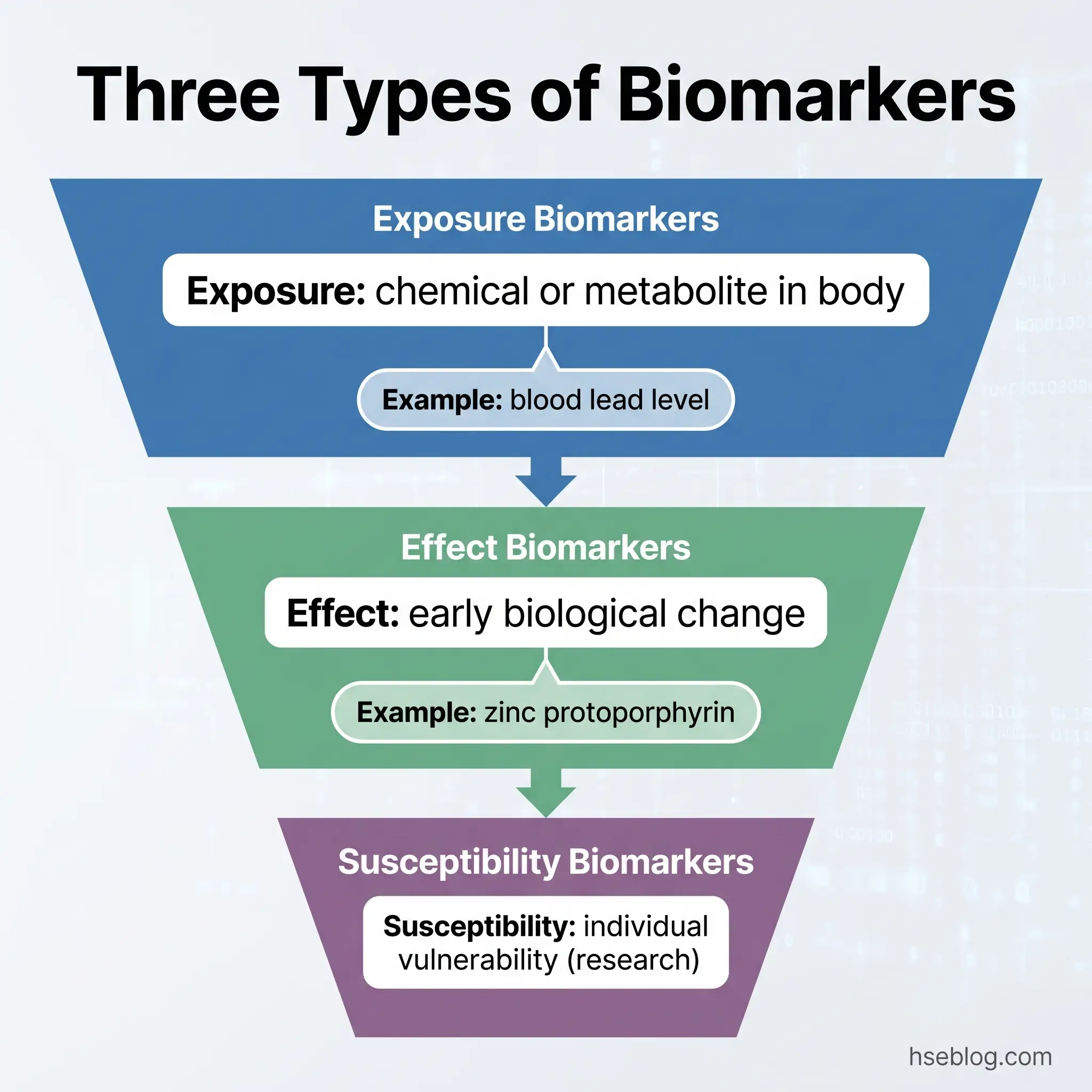
The choice of what to measure in a worker’s biological specimen is not arbitrary. It depends on the chemical’s behaviour inside the body — how it is absorbed, metabolised, distributed, and eliminated. Three categories of biomarkers are recognised, each serving a different purpose in exposure assessment.
Biomarkers of Exposure
Biomarkers of exposure are the most commonly used in routine occupational biological monitoring. They measure the chemical itself or its metabolite in a body fluid, confirming that absorption has occurred and quantifying the internal dose.
The chemical–biomarker pairs that practitioners encounter most frequently include:
- Lead → blood lead level (BLL): The reference biomarker for lead exposure worldwide. Lead accumulates in blood, soft tissue, and bone, with a blood half-life of approximately 30 days. Blood lead reflects recent and ongoing exposure.
- Cadmium → cadmium in blood and cadmium in urine: Blood cadmium reflects recent exposure; urinary cadmium reflects cumulative body burden over years, because cadmium accumulates in the renal cortex with a biological half-life exceeding 10 years.
- Benzene → urinary phenol and urinary S-phenylmercapturic acid (S-PMA): S-PMA is the more specific metabolite; urinary phenol can be influenced by non-occupational sources.
- Isocyanates → urinary diamines (e.g., MDA for MDI, TDA for TDI): Critical for assessing dermal absorption, since isocyanate exposure frequently occurs through skin contact with uncured product.
- Organophosphate pesticides → red blood cell cholinesterase and plasma cholinesterase: Cholinesterase depression indicates absorption, though this sits on the boundary between exposure and effect biomarkers.
Biomarkers of Effect
Biomarkers of effect measure early, reversible changes in the body resulting from chemical absorption. They do not measure the chemical itself but rather its biological consequence. Zinc protoporphyrin (ZPP) in blood, for instance, indicates that lead has interfered with the haem synthesis pathway — it is a marker of lead’s effect on haemoglobin production, not a measure of lead concentration itself. Similarly, beta-2-microglobulin in urine serves as an early indicator of cadmium-induced kidney tubular damage.
These markers are clinically significant because they bridge the gap between exposure assessment and health impact. A biomarker of exposure tells you the substance entered the body; a biomarker of effect tells you the body is responding to it. In programme design, both are valuable — but effect biomarkers carry greater urgency, because an elevated effect marker suggests that current controls are not preventing biological harm.
Biomarkers of Susceptibility
Biomarkers of susceptibility identify genetic or acquired characteristics that make certain individuals more vulnerable to a specific chemical’s toxic effects. ALAD polymorphisms affecting individual sensitivity to lead toxicity are the most-cited example. These biomarkers are primarily used in research settings and are not part of routine occupational monitoring. Their inclusion here is for completeness — practitioners should be aware they exist but will not typically encounter them in programme design.
The choice of biological matrix — blood, urine, or exhaled breath — depends on the chemical’s toxicokinetics. Substances with short biological half-lives, such as many solvents, require precise sampling timing: end of shift, end of the work week. A urine sample collected Monday morning will not capture Friday’s toluene exposure because the metabolites have already been excreted. Substances that accumulate, such as cadmium, are less timing-sensitive because the body burden changes slowly relative to any single shift.
When Is Biological Monitoring Needed?
This is the decision that most practitioners get to last, when it should come first. Biological monitoring is too often treated as a fallback measure — deployed after an incident, a health complaint, or an enforcement visit — rather than as a proactive tool built into the exposure assessment from the start. The triggers for implementing it fall into two categories: regulatory mandates (where the law requires it) and professional-practice triggers (where competent occupational hygiene demands it even without a specific legal instruction).
Six conditions should prompt the decision to implement biological monitoring:
- The chemical carries a skin notation. If the substance’s occupational exposure limit table entry includes a skin notation (Sk, skin, or equivalent), dermal absorption is a recognised significant exposure route. Air monitoring alone will systematically underestimate the true absorbed dose. Biological monitoring is the only reliable method for assessing total uptake. Common examples include isocyanates, organophosphate pesticides, some glycol ethers, and certain aromatic amines.
- PPE effectiveness needs verification. The gap between a respirator’s assigned protection factor in a laboratory test and its actual performance across an eight-hour shift on a moving worker is well documented. Biological monitoring provides the only direct evidence of whether the PPE programme is achieving the internal-dose reduction it was designed for.
- Exposure is variable or unpredictable. Routine air monitoring captures steady-state conditions. Maintenance shutdowns, process upsets, emergency interventions, and non-routine tasks generate exposure spikes that a scheduled air sampling programme may miss entirely. Biological monitoring integrates all exposure over the sampling period, including the spikes.
- Multiple exposure routes operate simultaneously. A worker in a chromium plating facility may inhale chromium (VI) mist, absorb it through skin contact with plating solutions, and ingest trace amounts from contaminated break areas. Air monitoring captures one of those three pathways. Biological monitoring — urinary chromium in this case — captures the combined result.
- Health surveillance findings suggest possible overexposure. When clinical assessment or biological effect monitoring identifies early health changes in workers whose air monitoring results appear satisfactory, the discrepancy itself is the trigger. Something in the exposure assessment is missing, and biological monitoring is the tool that finds it.
- The relevant regulation mandates it. Specific jurisdictional requirements are detailed below.
Watch For: The most common organisational failure is reasoning backwards — concluding that because air monitoring results are within limits, biological monitoring is unnecessary. That reasoning is circular. If a chemical has a skin notation, the air result alone cannot establish adequate control, regardless of its magnitude.
Regulatory Triggers by Jurisdiction
Requirements for biological monitoring differ substantially between jurisdictions. An HSE professional operating across borders — or advising a multinational operation — needs to know which chemicals trigger mandatory monitoring under which framework. This is the section where jurisdictional precision matters most.
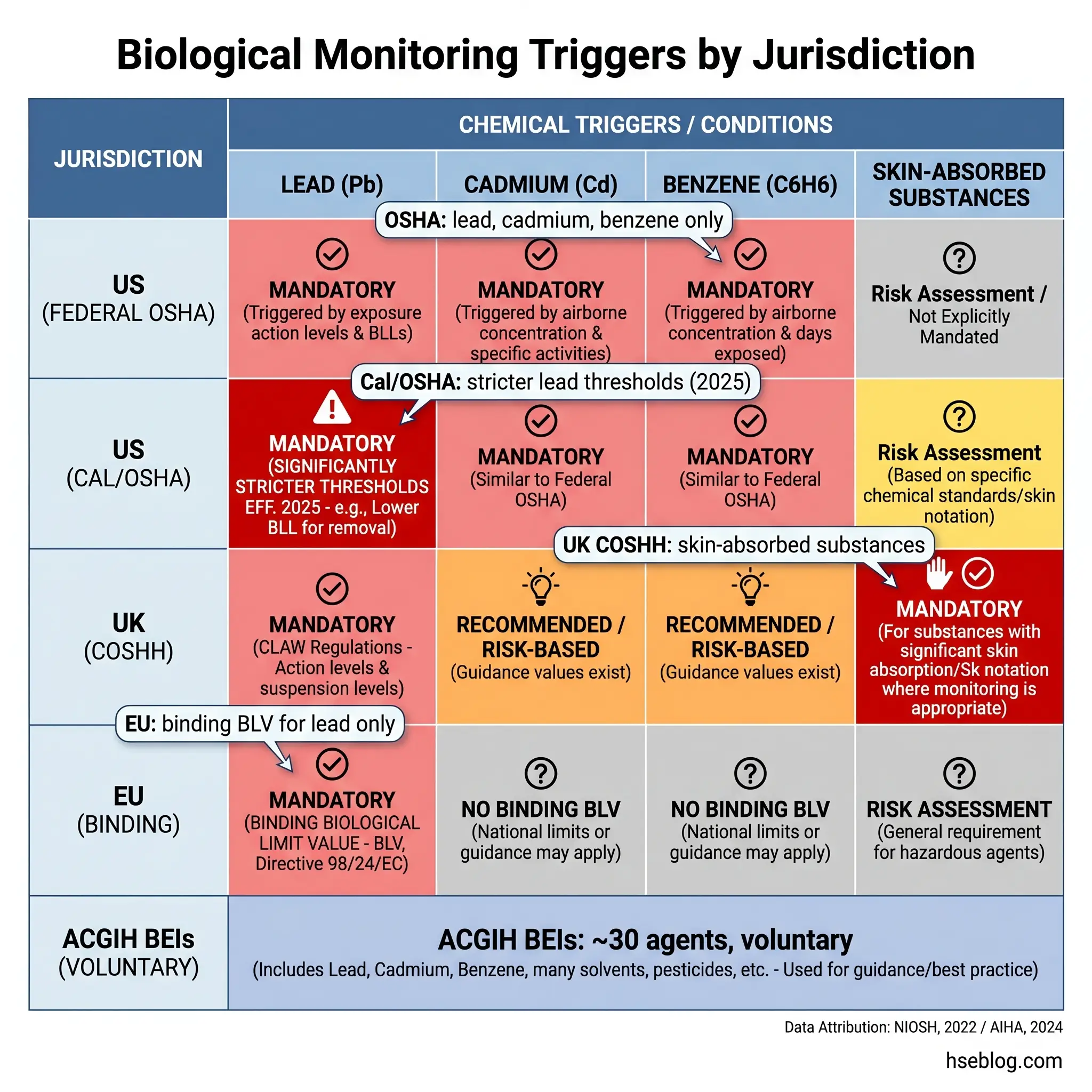
United States — Federal OSHA
Federal OSHA mandates biological monitoring for three substances only. Under 29 CFR 1910.1025 (general industry) and 1926.62 (construction), employers must provide blood lead level and zinc protoporphyrin testing for all workers exposed above the action level of 30 µg/m³ for more than 30 days per year. Monitoring frequency is every six months at minimum, increasing to every two months if the BLL reaches or exceeds 40 µg/dL. Medical removal is triggered at a BLL of 50 µg/dL in general industry.
Under 29 CFR 1910.1027, cadmium monitoring requires periodic measurement of cadmium in blood, cadmium in urine, and beta-2-microglobulin in urine.
Under 29 CFR 1910.1028, benzene biological monitoring (urinary phenol) is required following an unplanned release, with end-of-shift and 72-hour follow-up specimens.
United States — Cal/OSHA (California)
Cal/OSHA approved revised lead standards effective January 1, 2025, reducing the permissible exposure limit from 50 µg/m³ to 10 µg/m³ and the action level from 30 µg/m³ to 2 µg/m³ (AIHA, 2024). The practical effect is dramatic: biological monitoring requirements now trigger at far lower exposure levels, and the target is maintaining blood lead levels below 10 µg/dL — aligning with the current CDC reference value for adults (CDC/NIOSH, 2024), rather than the federal OSHA standard’s 1978-era thresholds. Any site operating under California jurisdiction must use these stricter values as the reference.
Jurisdiction Note: OSHA’s federal medical removal trigger for lead remains at 50 µg/dL — set in 1978. The CDC reference BLL of concern for adults is now 10 µg/dL (CDC/NIOSH, 2024). Cal/OSHA’s 2025 standard aligns with the CDC reference. ACOEM and CDPH recommend medical removal at two consecutive BLLs between 20–29 µg/dL. Always apply the stricter value applicable to your jurisdiction.
United Kingdom — COSHH
COSHH Regulation 10 (UK, 2002, amended 2005) requires exposure monitoring where necessary to maintain adequate control. For substances where skin absorption makes air monitoring insufficient, biological monitoring becomes the appropriate assessment tool. HSE publishes Biological Monitoring Guidance Values (BMGVs) in EH40 Table 2 — these are not legally binding exposure limits but represent concentrations at which control should be reviewed. Separate mandatory biological monitoring requirements exist under the Control of Lead at Work Regulations.
European Union
EU Directive 98/24/EC provides the legal basis for binding biological limit values (BLVs) and indicative biological guidance values (BGVs) across member states. Lead is currently the only substance with a binding EU BLV. Directive 2004/37/EC, covering carcinogens and mutagens at work (last amended 2022), requires that health surveillance include biological surveillance “where appropriate” — language that places the judgement on the competent person conducting the risk assessment.
ACGIH — Biological Exposure Indices (International Reference)
ACGIH publishes BEIs for approximately 30 chemical agents (NIOSH, 2022). These are voluntary guidance values representing biomarker concentrations expected in specimens from healthy workers experiencing inhalation exposure at the Threshold Limit Value. BEIs are not legally enforceable — ACGIH explicitly states they should not be adopted as legal standards without additional risk-management analysis. Despite this, they are the most widely referenced set of biological guidance values internationally and serve as the professional-practice benchmark for chemicals not covered by jurisdiction-specific mandates.
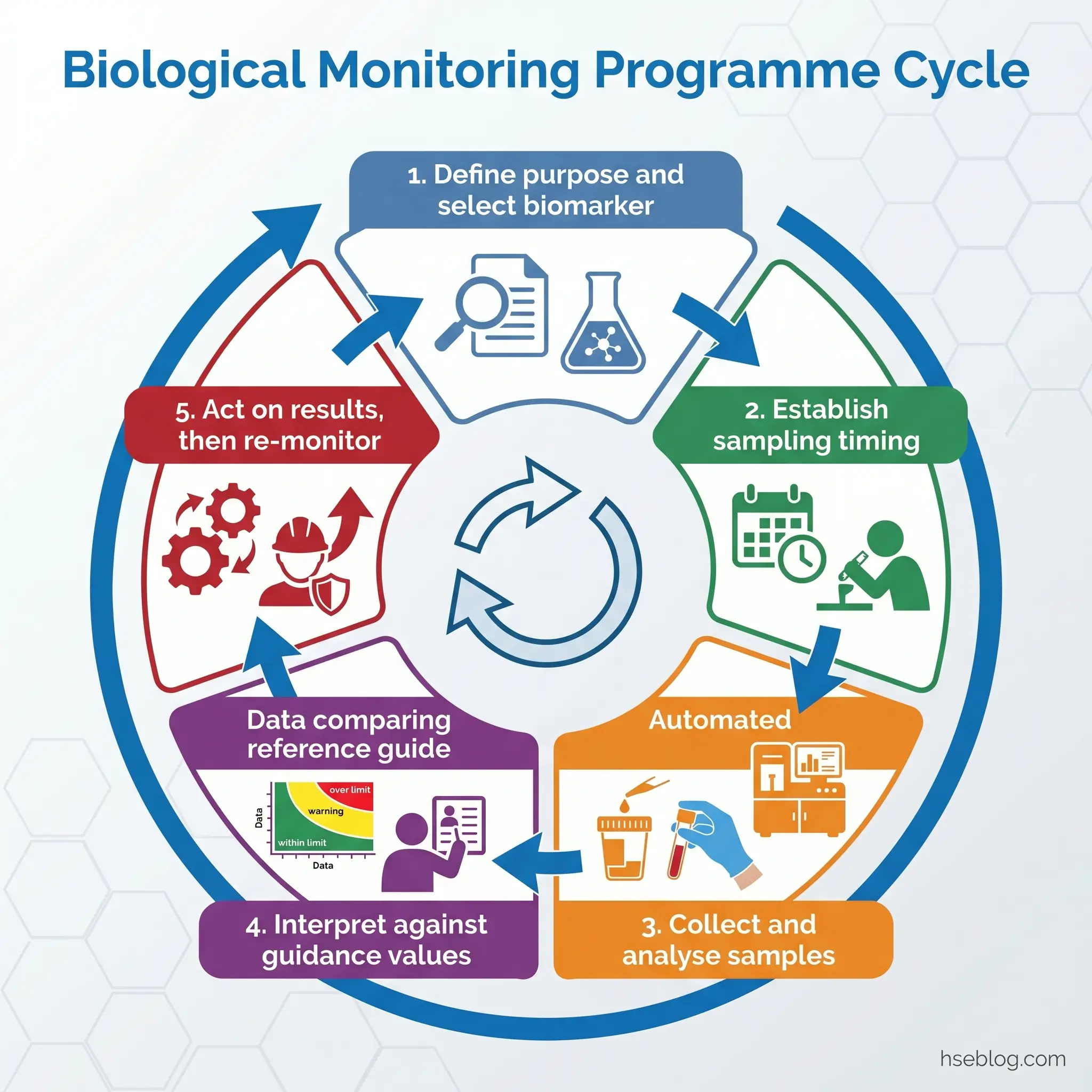
How a Biological Monitoring Programme Works
A biological monitoring programme is not a sampling schedule. It is a managed system that begins with a defined purpose and ends with action taken on results. The most common programme failure — consistent across published reviews — is not analytical error or sample contamination. It is the absence of a pre-defined action pathway for results that exceed reference values. Samples are collected, analysed, and filed. Nobody decided, before the first sample was drawn, what would happen if a worker’s result came back above the guidance value.
The EU-OSHA guide on biological monitoring at work, published in 2025, provides the most current framework for programme design across EU member states. Its principles apply broadly to any jurisdiction. The programme lifecycle follows a logical sequence, and each step depends on the one before it.
Define the programme purpose. Is this routine surveillance of an ongoing exposure? Post-incident assessment after a release? Verification that a newly installed engineering control is achieving the intended dose reduction? The purpose determines the biomarker selection, sampling frequency, and interpretation framework.
Appoint a competent person. Biological monitoring programme design and result interpretation require specific expertise — an occupational hygienist, occupational health physician, or nurse specialist with training in toxicokinetics and exposure assessment. This is not a task for the general safety officer to absorb into an already full workload.
Select the appropriate biomarker and matrix. This decision flows from the chemical’s toxicokinetics. A substance with a biological half-life of hours (many solvents) requires end-of-shift sampling, often at the end of the working week to capture weekly accumulation. A substance with a half-life of years (cadmium) is relatively timing-insensitive. The NIOSH guidance on applying biological monitoring methods provides detailed selection criteria for common chemical–biomarker–matrix combinations.
Establish sampling timing and frequency. Timing is the single most frequent source of error in biological monitoring programmes. A urine sample for hippuric acid (toluene metabolite) collected on Monday morning tells you nothing about the previous week’s exposure — the metabolite has been excreted over the weekend. Regulatory schedules set minimum frequencies (OSHA’s lead standard requires six-monthly minimum), but the competent person should set the actual frequency based on the chemical’s kinetics and the programme’s purpose.
Implement collection, handling, and chain-of-custody procedures. Biological samples are subject to the same rigour as any analytical specimen. Contamination at collection — a blood sample drawn near a dusty work area, a urine sample collected without prior handwashing — invalidates the result. Chain-of-custody documentation protects the programme’s legal standing.
Use an accredited analytical laboratory. Results are only as reliable as the analytical method. Laboratories should be accredited for the specific biomarker being measured, not simply accredited in general terms.
Interpret results against the relevant guidance value. Which guidance value applies depends on jurisdiction and chemical. BMGVs in the UK (from EH40 Workplace Exposure Limits including BMGVs), BEIs internationally, BATs in Germany, BLVs in the EU where they exist. The critical practitioner judgement: a single result above a guidance value is not necessarily cause for alarm, but it is always cause for investigation. Trending — tracking individual and group results over time — is more informative than any single data point.
Act on results. This is where programmes succeed or fail. An exceedance triggers a structured response: verify the result (repeat sampling if needed), investigate the exposure source, review engineering controls, assess PPE adequacy, review work practices, and re-monitor after corrective action. The programme plan must define these decision rules before the first sample is collected, not after the first exceedance arrives.
Audit Point: When reviewing a biological monitoring programme, the first question is not “are you sampling on schedule?” but “what is your documented action pathway for a result above the guidance value?” If that pathway does not exist in writing before the programme starts, the programme has a structural deficiency.
Interpreting Biological Monitoring Results
A result above a BMGV, BEI, or equivalent guidance value does not mean the worker is ill. This distinction is critical and frequently misunderstood — by employers, by workers, and sometimes by the professionals running the programme.
What an exceedance means: the amount of chemical absorbed by this worker, through all routes combined, exceeds the level that the guidance value was designed to flag. The appropriate response is to investigate why — which exposure pathway is responsible, which control has failed, which work practice has changed — and to correct the source.
What an exceedance does not mean: a clinical diagnosis, a determination of impairment, or grounds for disciplinary action against the worker. The result is information about the control system, not about the worker’s performance.
Equally, a result within normal range does not guarantee that the individual worker is free from risk. Biological monitoring results are influenced by individual variability in metabolism, hydration, diet (certain foods can elevate urinary metabolites that overlap with occupational biomarkers), smoking status, concurrent medication, and the precise timing of sample collection relative to exposure. A single data point is a snapshot. The value of biological monitoring increases with repeated measurements, trend analysis, and group comparisons.
Ethical and Legal Considerations
Biological monitoring requires taking a physical sample from a worker’s body. That act carries ethical obligations that go beyond standard workplace health and safety procedure, and the organisations that handle these obligations poorly tend to fail in a specific, predictable way: not at the consent stage, but at the communication stage.
Informed consent is the starting point. Workers must understand what is being measured, why, what the results mean, who will see them, and what happens if results are abnormal — before they provide a sample. Consent must be genuine and documented. The exception to voluntariness is narrow: where specific regulations mandate biological monitoring participation (the Control of Lead at Work Regulations in the UK, for instance, or OSHA’s lead standard in the US where the employer must make monitoring available and the worker’s refusal carries specific documented consequences).
Confidentiality is the second obligation. Individual biological monitoring results are medical data. In EU and UK jurisdictions, GDPR and UK data protection legislation classify biological sample results as special-category personal data, subject to the highest tier of data protection obligations. The occupational health professional holds individual results confidentially. What the employer may see — and all the employer needs to see — is whether the result is above or below the action level, and what group-level trends indicate about control effectiveness.
Workers’ right to know is the third. Every worker whose biological sample is taken has the right to be informed of their own result and, critically, what that result means in plain language. The failure mode encountered most consistently across programme reviews is communication that stops at a letter: “Your result was within the normal range” or “Your result was above the guidance value — please contact occupational health.” No explanation of what the number represents. No context for what “above the guidance value” means for them personally. No named contact. Effective programmes invest as much effort in result communication — clear, individual, accessible — as in sample collection logistics.
Prohibition on misuse of results is the fourth. Biological monitoring results exist to protect health, not to make employment decisions unrelated to health protection. Using a result to terminate, demote, or reassign a worker for reasons other than health-protective action violates the purpose of the programme and, in most jurisdictions, the law.
The Fix That Works: Before collecting the first sample, write the result communication template. Decide who delivers results, in what format, with what explanatory context, and with what follow-up pathway. If the communication plan does not exist before sampling begins, the programme will default to the impersonal letter — and worker trust, which is the foundation of voluntary participation, erodes.
What Are Biological Monitoring Guidance Values?
Guidance values are the reference points against which biological monitoring results are interpreted. Without them, a blood lead level of 15 µg/dL is just a number. With the appropriate guidance value, it becomes information — either confirming adequate control or flagging a need for investigation.
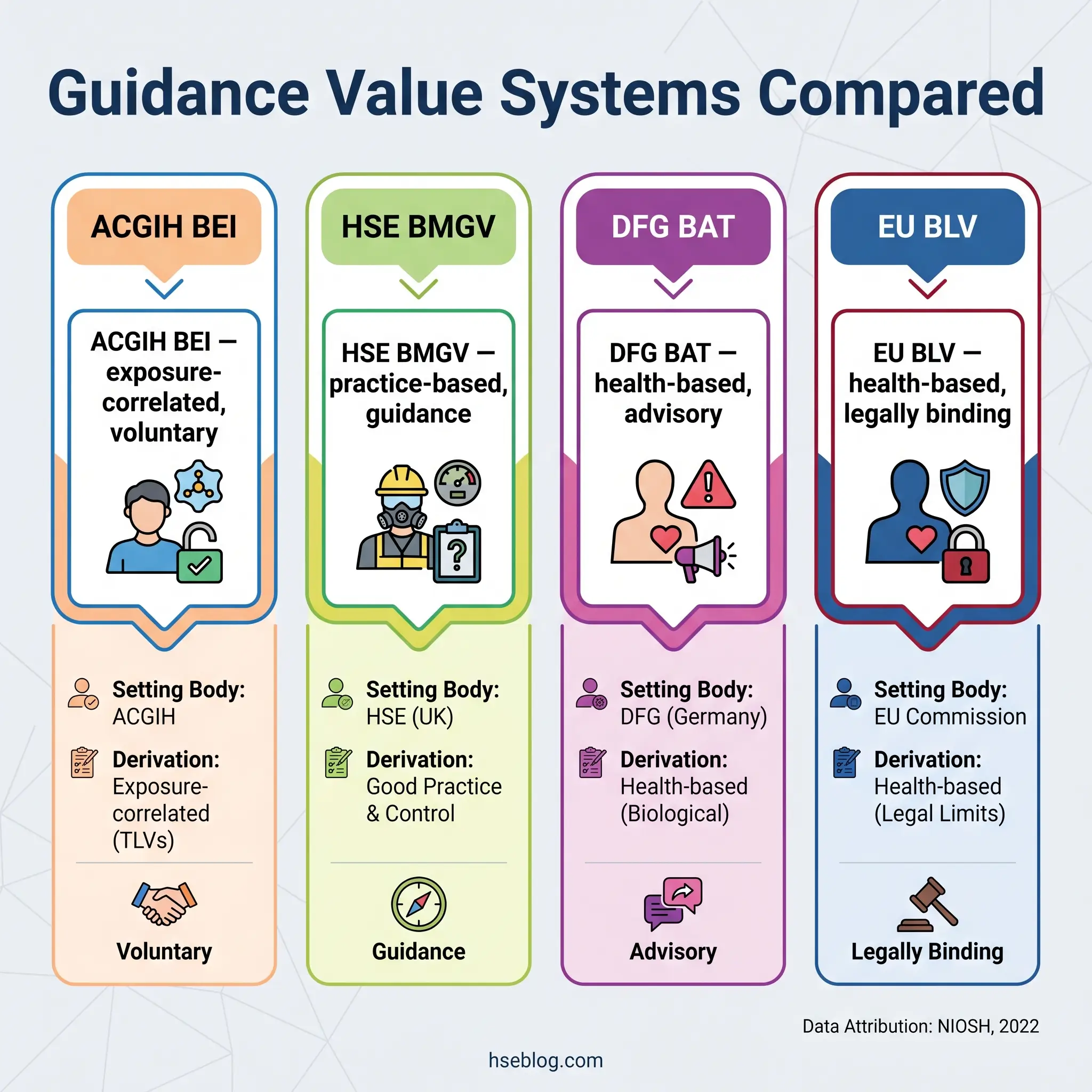
Several systems exist, set by different organisations, using different derivation methods, carrying different legal weight. The critical practitioner insight: these systems are not interchangeable, and their thresholds may differ for the same chemical.
ACGIH Biological Exposure Indices (BEIs) are published annually and cover approximately 30 chemical agents (NIOSH, 2022). Each BEI represents the biomarker concentration expected in specimens from healthy workers experiencing inhalation exposure at the corresponding Threshold Limit Value. BEIs are exposure-correlated — derived from the relationship between airborne concentration and biological level — rather than directly health-based. ACGIH explicitly states that BEIs are guidance values, not legal standards, and should not be adopted as enforceable limits without additional analysis. Despite this caveat, BEIs serve as the most widely used professional-practice reference internationally.
HSE UK Biological Monitoring Guidance Values (BMGVs) are published in EH40 Table 2. BMGVs are set at levels achievable when good occupational hygiene practice is applied. They are not health-based limits in the strict toxicological sense — a result at or below the BMGV does not guarantee safety, and a result above it does not confirm harm. Their legal status is as guidance within the COSHH framework: an exceedance obligates investigation and control review, not automatic medical removal.
German DFG Biologische Arbeitsstoff-Toleranz-Werte (BATs) are biological tolerance values derived by the Deutsche Forschungsgemeinschaft. These are health-based — set at concentrations below which adverse health effects are not expected in healthy workers with repeated exposure. Germany also publishes Expositionsäquivalente für krebserzeugende Arbeitsstoffe (EKAs) for carcinogenic substances.
EU Binding Biological Limit Values (BLVs) exist under Directive 98/24/EC but have been established for only one substance — lead. Indicative Biological Guidance Values (BGVs), based on background population levels in unexposed individuals, are a more recent development intended to complement BLVs. The practical implication: EU member states may set national biological limit values that are stricter than the EU BLV, but not less protective.
Only 60–90 chemicals have validated biomonitoring methodologies out of the thousands of chemicals used in workplaces (EU-OSHA OSHwiki, 2011). That limitation defines the current boundary of biological monitoring as a tool — for most workplace chemicals, no validated biomarker or analytical method exists, and air monitoring remains the primary assessment approach.
Frequently Asked Questions
Conclusion
The fundamental lesson from the published record on biological monitoring is that the tool itself is well understood — the science of biomarkers, the analytical methods, the guidance value systems are mature. What consistently fails is the organisational architecture around the tool. Programmes collect samples without pre-defined action pathways for exceedances. Air monitoring results are accepted as complete exposure assessments for chemicals that carry skin notations. Individual results are communicated through impersonal letters that leave workers uninformed about what their own data means. The highest-impact change most organisations can make is not adding more chemicals to the monitoring list — it is building the decision framework before drawing the first sample.
The regulatory landscape is shifting toward stricter thresholds and broader application. Cal/OSHA’s 2025 revised lead standard — reducing the PEL by 80% and the action level by 93% (AIHA, 2024) — signals the direction. The EU-OSHA 2025 biomonitoring guide establishes common principles across member states for the first time. For practitioners, the professional-practice question is no longer whether biological monitoring is worth the investment but whether the programme behind it is designed to actually protect people — not just generate data.
Biological monitoring measures what entered the body. Whether that measurement protects anyone depends entirely on what happens after the result is reported.