TL;DR — Key Numbers
- ~799,000 occupational cancer deaths globally per year — up from 666,000 a decade earlier, making workplace carcinogen exposure one of the largest occupational health burdens worldwide (ILO/ICOH, 2022).
- 2–8% of all cancers are caused by workplace carcinogenic exposures, with the true fraction likely higher when probable carcinogens and synergistic effects are included (NIOSH/CDC, 2024).
- 120,000 new work-related cancer cases annually in the EU alone — cancer accounts for nearly half of all occupational disease deaths across EU member states (EU-OSHA).
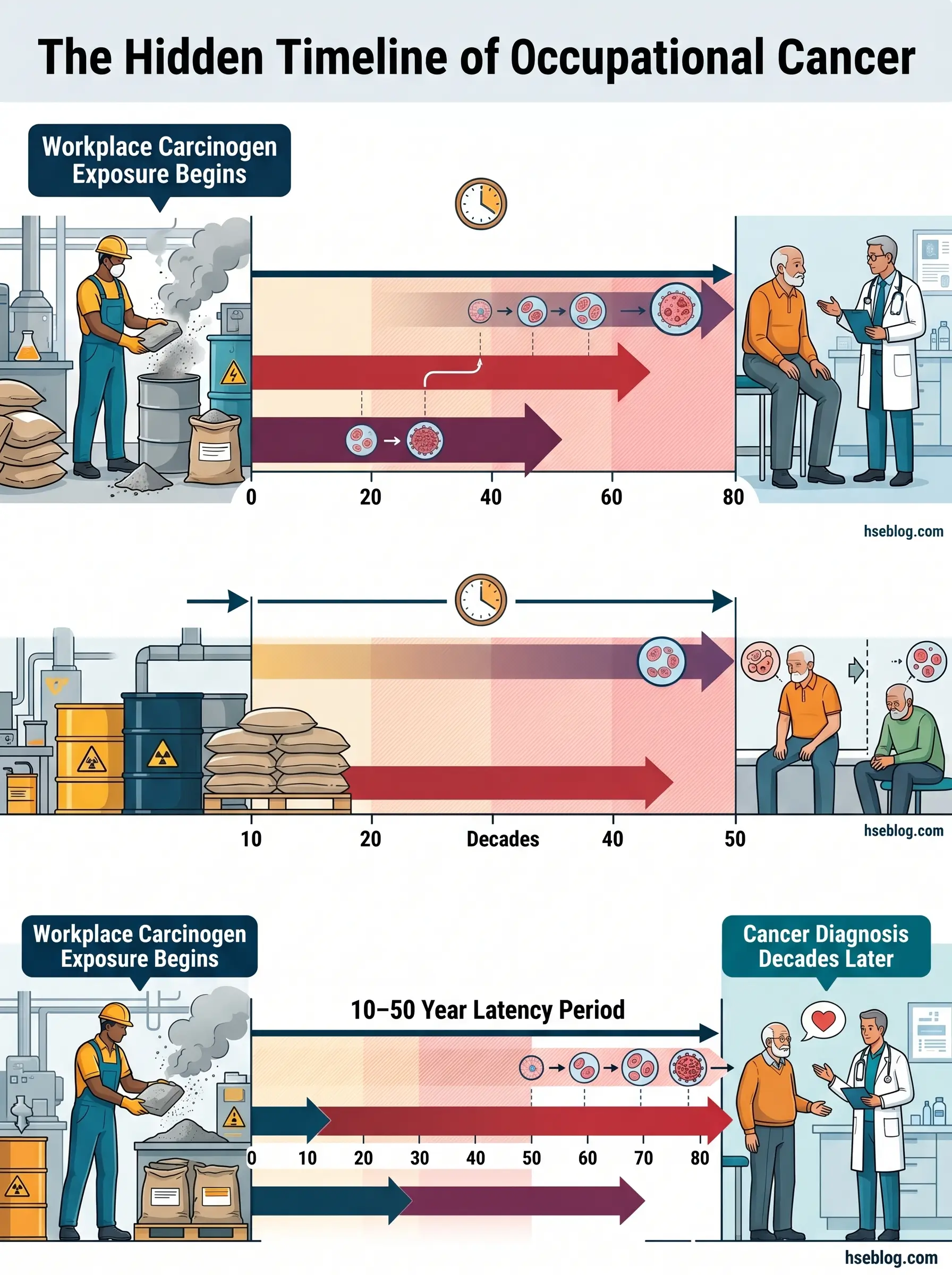
- 10–50 year latency periods mean that today’s diagnoses reflect exposures from decades past, while today’s uncontrolled exposures will manifest in workers who are currently healthy.
Occupational cancer is cancer caused wholly or partly by exposure to carcinogenic substances, processes, or conditions at work. Common types include lung cancer, mesothelioma, and bladder cancer. With latency periods often exceeding 10–40 years, today’s diagnoses reflect past workplace conditions. An estimated 2–8% of all cancers globally are attributable to occupational exposures (NIOSH/CDC, 2024), and most are preventable through elimination, substitution, and engineering controls.
What Is Occupational Cancer and Why Does It Matter?
In 2023, 2,218 people died from mesothelioma in Great Britain — every one of them from asbestos fibres inhaled decades earlier, in workplaces that may no longer exist (UK HSE, 2025). That single statistic captures the defining paradox of occupational cancer: the harm is invisible at the point of exposure, and lethal by the time it becomes visible.
Occupational cancer differs from general cancer in one critical respect: the causal or contributory exposure arises directly from work. The latency period — the gap between first exposure and clinical diagnosis — typically spans 10 to 50 years depending on the carcinogen and cancer type. Mesothelioma from asbestos averages 30–40 years. Bladder cancer from aromatic amines may surface 15–25 years after exposure ends. This means that workers diagnosed today were exposed under working conditions from the 1980s, 1990s, or early 2000s — and that workers being exposed today under inadequate controls will develop cancers in the 2040s and 2050s.
The scale of under-recognition compounds the problem. In developing economies where cancer registries rarely capture occupational history data, the link between a worker’s diagnosis and their past exposure is often never made. Even in jurisdictions with mature reporting systems, occupational cancer remains under-reported because the latency period severs the temporal connection between cause and effect. An estimated 2–8% of all cancers worldwide are caused by workplace carcinogenic exposures (NIOSH/CDC, 2024), but some researchers argue the fraction is higher for specific cancer types when Group 2A probable carcinogens and unrecognised synergistic exposures are included.
For HSE professionals, this creates an asymmetry in perceived urgency that is the central challenge. Safety managers focus disproportionately on acute hazards — falls, machine guarding, electrical contact — because the consequence is immediate and the causal chain is obvious. Carcinogen exposure control receives less operational urgency precisely because harm is invisible for decades. Reviewing the published incident record, the pattern is clear: organisations that invest heavily in acute hazard prevention while neglecting chronic exposure controls are not making a risk-based decision. They are making a visibility-based one.
How Many Workers Are Affected? The Global Burden of Occupational Cancer
The numbers are large enough that they resist intuition. Globally, an estimated 799,000 workers die from occupational cancer each year — a figure that has risen from approximately 666,000 a decade earlier, reflecting both improved surveillance and an expanding exposed workforce in industrialising economies (ILO/ICOH, 2022). In 2019, 2.93 million workers died from work-related accidents and diseases worldwide, with 2.58 million of those deaths attributable to occupational disease rather than acute injury (ILO/ICOH, 2024). Cancer is one of the leading disease categories within that total, alongside cardiovascular and respiratory conditions.
Regional data sharpens the picture. In the European Union, approximately 120,000 new work-related cancer cases occur annually (EU-OSHA), and cancer accounts for nearly half of all occupational disease deaths across member states. In Great Britain, the HSE Cancer Burden Study estimated that occupational exposures account for approximately 5.3% of cancer deaths, translating to over 8,000 cancer deaths per year (Rushton et al., 2012). That study, based on 2005 baseline data, used conservative attribution methods and included only carcinogens with strong epidemiological evidence — the actual burden is almost certainly higher.
The gender dimension is evolving. Historically, men have borne the greater occupational cancer burden because of their concentration in mining, construction, and heavy manufacturing. However, research increasingly recognises cancer risks in female-concentrated sectors — healthcare workers handling cytotoxic drugs, hairdressers exposed to chemical mixtures, and agricultural workers with pesticide and solar UV exposure. The historical research gap means that attributable fractions for women’s occupational cancers are likely underestimated.
Watch For: Burden estimates are inherently conservative. They typically count only IARC Group 1 agents with established dose-response data. The true occupational cancer toll — including probable carcinogens, mixed exposures, and unrecognised synergistic effects — almost certainly exceeds published figures.
What Are the Main Occupational Carcinogens and How Are They Classified?
Understanding which substances cause occupational cancer requires distinguishing between hazard classification — the scientific determination that an agent can cause cancer — and risk assessment — the evaluation of whether it will cause cancer under specific workplace exposure conditions. The most authoritative hazard classification system is the IARC Monographs programme, operated by the World Health Organization’s International Agency for Research on Cancer.
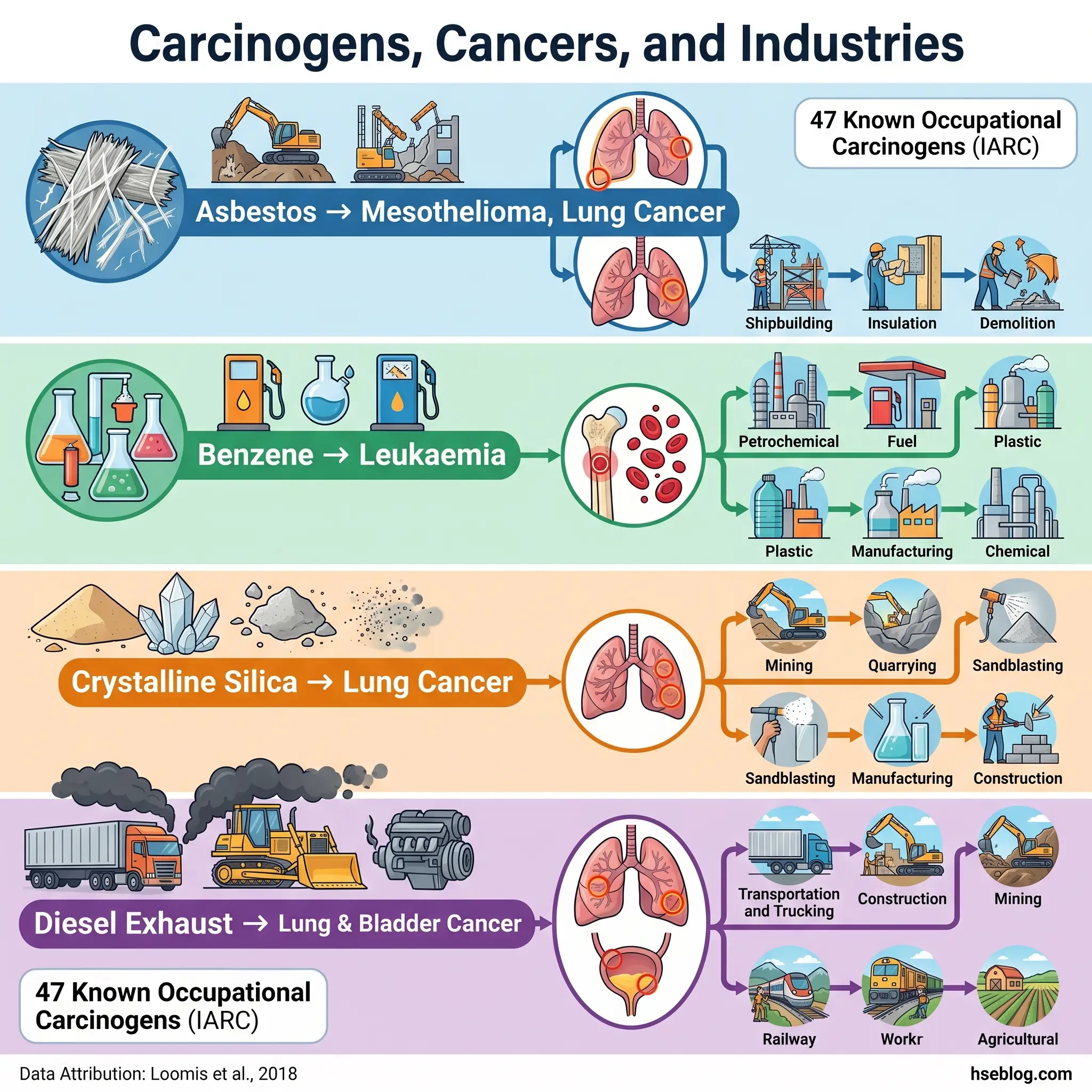
IARC classifies agents into four groups based on the strength of scientific evidence. Group 1 agents are carcinogenic to humans, supported by sufficient evidence from epidemiological studies. Group 2A agents are probably carcinogenic, with limited evidence in humans but sufficient evidence in animal studies combined with strong mechanistic data. Group 2B agents are possibly carcinogenic, and Group 3 agents are not classifiable due to inadequate evidence. As of the most recent published analysis, 47 agents have been identified as known occupational carcinogens — Group 1 agents with sufficient evidence specifically from worker studies — up from 28 in 2004 (Loomis et al., 2018). The full IARC classified list of carcinogenic agents is regularly updated as new Monograph volumes are published.
A common misconception among practitioners is treating IARC classification as a direct measure of workplace risk. It is not. A Group 1 carcinogen present at trace concentrations with minimal worker contact may pose lower practical risk than a Group 2A agent to which workers are exposed at high concentrations over prolonged periods. Risk assessment must account for actual exposure conditions — concentration, duration, frequency, route of entry, and individual susceptibility — not just hazard classification alone.
Major occupational carcinogens fall into several categories. Chemical substances include asbestos (all fibre types), benzene, formaldehyde, crystalline silica, hexavalent chromium, and vinyl chloride. Physical agents include ionising radiation and ultraviolet radiation. Biological agents include hepatitis B and C viruses and human papillomavirus in healthcare settings. Process-generated substances — those created during work rather than deliberately used — include diesel engine exhaust, welding fumes, polycyclic aromatic hydrocarbons (PAHs), and rubber manufacturing fumes. IARC also classified night shift work as Group 2A (probably carcinogenic) based on evidence linking circadian disruption to breast cancer risk.
In the US, OSHA’s Laboratory Standard (29 CFR 1910.1450, US) defines “select carcinogens” as substances classified by NTP as “known to be human carcinogens,” by IARC as Group 1, or by IARC as Group 2A/2B with NTP “reasonably anticipated” classification. This definition governs how laboratory employers must handle these substances in Chemical Hygiene Plans.
Key Carcinogen-to-Cancer-Site Associations
The following associations represent the most established links in occupational epidemiology. Practitioners conducting workplace risk assessments should use these as a starting reference when identifying which cancer outcomes are relevant to their site’s specific exposures:
| Carcinogen | Primary Cancer Site(s) | Key Exposure Industries |
|---|---|---|
| Asbestos (all forms) | Mesothelioma, lung cancer | Construction, shipbuilding, insulation, demolition |
| Benzene | Leukaemia (AML) | Petrochemical, rubber, printing |
| Crystalline silica | Lung cancer | Construction, mining, quarrying, foundries |
| Hexavalent chromium | Lung cancer, nasal cancer | Metal plating, stainless steel welding, pigment production |
| Formaldehyde | Nasopharyngeal cancer, leukaemia | Healthcare (pathology), wood products, chemical manufacturing |
| Wood dust | Nasal adenocarcinoma | Furniture, carpentry, sawmills |
| Vinyl chloride | Liver angiosarcoma | PVC manufacturing |
| UV radiation (solar) | Non-melanoma skin cancer, melanoma | Outdoor workers across all sectors |
| Diesel engine exhaust | Lung cancer, bladder cancer | Mining, transport, construction |
Which Industries and Occupations Carry the Highest Risk?
The published record on occupational cancer shows a consistent pattern: risk concentrates in sectors where workers encounter carcinogenic substances as routine byproducts of the work process itself, rather than as isolated incidents. The concentration is not accidental — it reflects the industrial processes that generate, release, or disturb carcinogens during normal operations.
Construction remains one of the highest-risk sectors globally. Workers face simultaneous exposure to multiple carcinogens — crystalline silica from concrete cutting and drilling, residual asbestos in pre-2000 buildings during refurbishment or demolition, diesel engine exhaust from heavy equipment, wood dust from carpentry operations, and solar UV radiation across all outdoor tasks. The combination of multiple concurrent carcinogenic exposures in construction makes single-substance risk assessment inadequate; cumulative and synergistic effects demand a broader exposure management approach.
Mining concentrates radon, uranium, crystalline silica, and diesel exhaust exposures in enclosed underground environments where ventilation constraints amplify airborne concentrations. Surface mining operations add solar UV to the exposure profile.
Chemical, rubber, and metal manufacturing expose workers to benzene, formaldehyde, hexavalent chromium, and PAHs through production processes. Foundry workers face crystalline silica from sand casting. Rubber manufacturing generates a complex mixture of carcinogenic compounds classified as Group 1 by IARC.
Firefighting was reclassified by IARC in 2022 as a Group 1 carcinogenic occupation — a significant shift from its previous 2B classification. Firefighters face exposure to combustion products including PAHs, benzene, formaldehyde, and particulates during structural fires, with dermal absorption increasingly recognised as a significant exposure route alongside inhalation.
Healthcare is an emerging risk area. The EU recognised this formally through Directive 2022/431, which expanded the Carcinogens, Mutagens and Reprotoxic Substances Directive to cover hazardous medicinal products (HMPs) — primarily cytotoxic drugs used in cancer treatment that are themselves carcinogenic. In early 2025, the European Commission published the definition and indicative list of HMPs containing CMR substances, operationalising protections for nurses, pharmacists, and other healthcare workers who prepare and administer these drugs.
Outdoor workers across all sectors face what is arguably the most widespread occupational carcinogen by exposed population: solar ultraviolet radiation. Non-melanoma skin cancer linked to occupational UV exposure is quantified in the WHO/ILO Joint Estimates as a substantial and growing burden, particularly as climate change increases UV intensity in many working latitudes.
The highest-risk workers are disproportionately found in small and medium enterprises. National burden studies consistently show that SMEs in construction and manufacturing account for elevated carcinogen exposure rates because they operate with older processes, limited occupational hygiene resources, and less systematic control programmes than large organisations with dedicated health and safety teams.
What Is the Regulatory Framework for Occupational Carcinogens?
The regulatory approach to occupational carcinogens varies significantly across jurisdictions — not just in the specific exposure limits set, but in the underlying philosophy governing how carcinogen exposure is managed.
United States (OSHA): The US framework relies on substance-specific standards under 29 CFR 1910.1001 through 1910.1053 (US), each setting Permissible Exposure Limits (PELs) for individual carcinogens with associated requirements for exposure monitoring, medical surveillance, and engineering controls. The 13 Carcinogens Standard (29 CFR 1910.1003, US) covers a group of named carcinogens without PELs — instead mandating engineering controls, regulated areas, and decontamination procedures to prevent any detectable exposure. The Hazard Communication Standard requires labelling and Safety Data Sheets for all carcinogenic substances.
European Union: The EU Carcinogens, Mutagens and Reprotoxic Substances Directive (Directive 2004/37/EC, EU) takes a substitution-first approach — employers must replace carcinogens with less hazardous alternatives unless they can demonstrate this is not technically possible. The Directive establishes binding Occupational Exposure Limits (OELs) for 41 CMR agents as of the most recent amendments (including Directives 2017/2398, 2019/130, 2019/983, 2022/431, and 2024/869). Individual health records for exposed workers must be maintained for 40 years after the last known exposure. In December 2025, the EU Council agreed its position on the 6th revision of the CMRD, adding welding fumes to Annex I and setting new OELs for cobalt compounds, PAHs, 1,4-dioxane, and isoprene — changes expected to prevent approximately 1,700 lung cancer cases over 40 years (Council of the EU, 2025).
United Kingdom: Under the COSHH Regulations 2002, Regulation 7(5) (UK), exposure to carcinogens and mutagens must be reduced to as low as reasonably practicable (ALARP). This is a stricter standard than simply meeting a numerical limit — employers must demonstrate that further reduction is not reasonably practicable before accepting any level of residual exposure. Workplace Exposure Limits carry specific “Carc” notations for carcinogenic substances.
International: The ILO List of Occupational Diseases includes 21 carcinogenic exposure agents for recognition by member states in developing national compensation and surveillance systems. The IARC Monographs programme (Volumes 1–141 as of 2025) provides the global scientific evidence base that underpins regulatory classification in every jurisdiction, though IARC classifications are advisory and not legally binding without national adoption.
Audit Point: A recurring failure pattern in compliance is treating the legal minimum as adequate cancer prevention. Many OSHA PELs for carcinogens have not been updated since the 1970s and are far less protective than current NIOSH Recommended Exposure Limits or ACGIH Threshold Limit Values. Practitioners operating to best practice — not just legal compliance — should benchmark against the most protective available standard.
Conflicting Exposure Limits: OSHA PEL vs NIOSH REL vs EU CMRD OEL
The divergence between regulatory limits for the same carcinogen across jurisdictions creates genuine operational complexity, particularly for multinational employers. Consider two of the most common occupational carcinogens:
Crystalline silica: OSHA’s PEL is 50 µg/m³ (8-hour TWA) (US). NIOSH’s REL is also 50 µg/m³ but carries a recommendation to keep exposures as low as feasible (US). The EU CMRD transitional OEL has been set at 0.05 mg/m³ (equivalent to 50 µg/m³) and is under review for possible further reduction (EU). While the numerical limits are currently aligned, the EU’s substitution-first legal obligation and the ALARP principle under UK COSHH impose a practical burden that goes well beyond meeting a number.
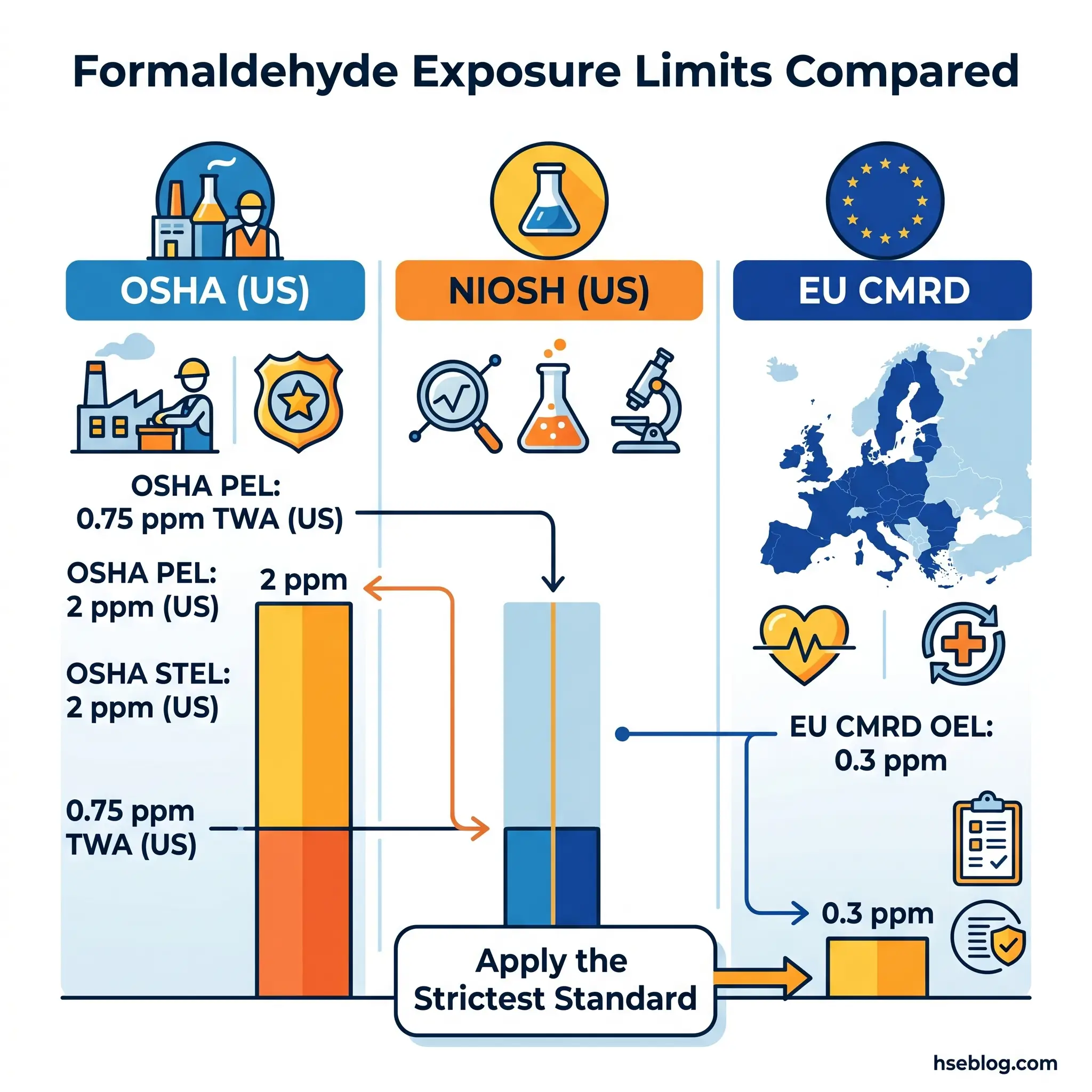
Formaldehyde: OSHA’s PEL is 0.75 ppm (8-hour TWA) with a 2 ppm short-term exposure limit (US). The EU CMRD OEL is 0.3 ppm, with a transitional period at 0.5 ppm for certain sectors extending to 5 April 2026 (EU). The EU limit is substantially stricter — an employer compliant with OSHA’s PEL could be in violation of EU requirements by a factor of more than two.
The practical judgement for practitioners is straightforward: when operating across jurisdictions or seeking best-practice protection, apply the most protective available standard. When operating in a single jurisdiction, meet the legal requirement but do not treat it as the target — the target is the lowest achievable exposure.
How Can Occupational Cancer Be Prevented? The Hierarchy of Controls Applied to Carcinogens
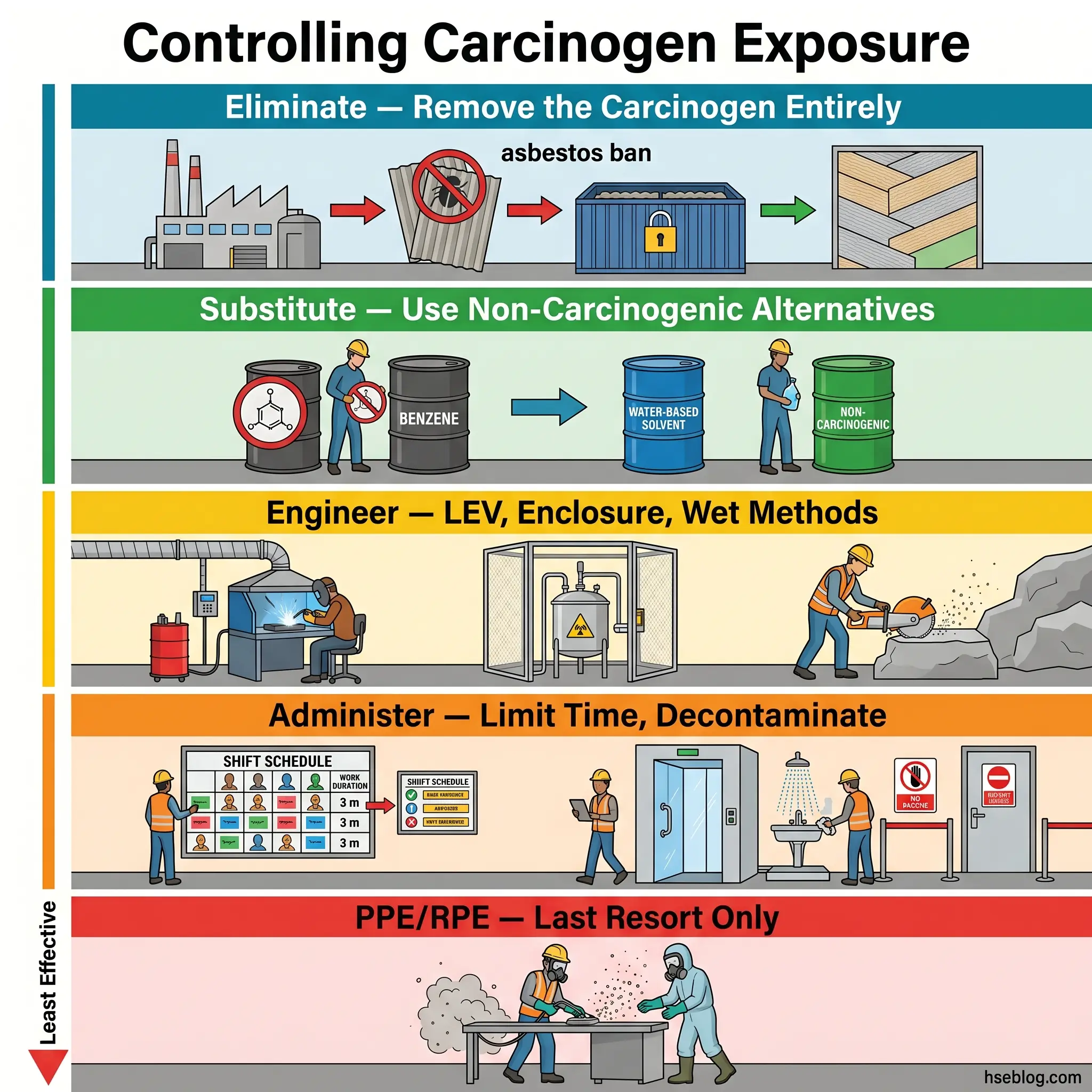
Prevention of occupational cancer follows the hierarchy of controls, but its application to carcinogens demands more rigour than for most workplace hazards. The non-threshold nature of many carcinogens — where no exposure level can be declared truly safe — means that every tier of control must be pushed as far as technically achievable, not merely to a numerical compliance point.
Elimination
Removing the carcinogenic substance or process from the workplace entirely is the only control that guarantees zero exposure. Asbestos bans — now in effect in over 60 countries — represent the most significant elimination success in occupational cancer history. The elimination of benzene as a general-purpose solvent in industries where alternatives exist is another landmark example. Elimination decisions are rarely simple, however; they require evaluating whether the function the carcinogen serves can be achieved through non-carcinogenic means without introducing unacceptable operational, quality, or safety trade-offs.
Substitution
Replacing a carcinogen with a less hazardous alternative is the next priority. The EU CMRD (Directive 2004/37/EC, EU) codifies substitution as a legal obligation — employers must substitute unless they demonstrate it is not technically possible. The practical challenge is what the toxicology literature calls “regrettable substitution”: replacing a known carcinogen with a poorly characterised substance that later proves equally or differently hazardous. Practitioners evaluating substitutes must ensure the replacement has been assessed for its own carcinogenic, mutagenic, and reprotoxic properties — not just for the specific cancer endpoint of the substance being replaced.
Engineering Controls
Where carcinogens cannot be eliminated or substituted, engineering controls must reduce exposure to the lowest achievable level. Enclosed systems prevent carcinogen release into the breathing zone. Local exhaust ventilation (LEV) captures airborne contaminants at the point of generation. Wet methods suppress dust for silica-generating processes. Automated handling removes workers from direct contact with carcinogenic materials.
The failure mode practitioners encounter most frequently with engineering controls is degradation over time. LEV systems lose effectiveness as ductwork accumulates deposits, filters become loaded, fan performance declines, and capture hoods are repositioned by workers who find them obstructive. Without scheduled maintenance, testing, and thorough examination — LEV examination is a legal requirement under COSHH (UK) at least every 14 months — engineering controls that were adequate at installation gradually become inadequate while appearing unchanged to untrained observation.
Administrative Controls
Administrative measures reduce exposure through work organisation rather than physical barriers. These include limiting time spent in exposure zones, rotating workers across tasks to distribute cumulative dose, restricting access to areas where carcinogens are present, and establishing decontamination protocols. Prohibition of eating, drinking, and smoking in exposure areas prevents ingestion and reduces hand-to-mouth transfer.
One administrative control that is critically under-implemented is “take-home” exposure prevention. Carcinogenic particles on work clothing, skin, and hair can be transferred to workers’ homes, potentially exposing family members. Known cases of mesothelioma in family members of asbestos workers who carried fibres home on their clothing illustrate the real consequences. Effective take-home prevention requires dedicated work clothing that stays on-site, on-site changing and showering facilities, and workplace laundering protocols — never allowing contaminated clothing to be taken home for domestic washing.
Personal Protective Equipment and Respiratory Protective Equipment
PPE and RPE are the last line of defence — used only to address residual risk that higher-tier controls cannot eliminate. For carcinogenic exposures, RPE selection must match the specific agent: particulate filters for dusts and fibres, combination cartridges for vapours, or supplied-air systems for environments where atmospheric contamination cannot be reliably controlled. Fit testing, training, and maintenance are non-negotiable requirements, not optional enhancements.
The most common prevention failure across the published record of occupational cancer investigations is over-reliance on RPE as a primary control rather than a last resort. RPE compliance degrades significantly during hot weather, physically demanding tasks, and extended shifts — exactly the conditions when exposure intensity is often highest. Effective carcinogen control programmes front-load engineering controls and use RPE only to manage the residual exposure that engineering and administrative measures cannot eliminate.
The Fix That Works: The EU’s STOP principle (Substitution, Technical measures, Organisational measures, Personal protection) mirrors the hierarchy of controls but codifies it as a legal sequence. Under the CMRD (EU), an employer who jumps directly to PPE without documenting why higher-tier controls are not reasonably practicable is non-compliant — not just operating below best practice.
What Role Does Health Surveillance Play in Occupational Cancer Prevention?
Health surveillance for carcinogen-exposed workers is one of the most critical — and most neglected — elements of occupational cancer prevention. Its purpose is not to prevent exposure; that is the function of the controls described above. Health surveillance exists to detect early biological effects, confirm that controls are working, and create a long-term record that connects individual workers to their specific exposure history.
Two distinct types of monitoring are involved. Exposure monitoring measures the level of carcinogen present in the work environment or the worker’s body. Workplace air sampling quantifies airborne concentrations. Biological monitoring measures carcinogen metabolites or biomarkers in blood, urine, or exhaled breath — for example, urinary metabolites of PAHs in coke oven workers, or blood benzene levels in petrochemical operators. Health surveillance, by contrast, monitors the worker’s health status through medical examinations, symptom screening, and long-term health outcome tracking. Both types work together: exposure monitoring confirms whether controls are containing the hazard, while health surveillance detects whether the hazard is nonetheless affecting the worker.
Legal requirements vary by jurisdiction but share common principles. OSHA’s substance-specific standards (US) mandate medical surveillance for workers exposed above action levels to regulated carcinogens — including periodic medical examinations, biological specimen collection, and worker notification. Under COSHH (UK), health surveillance is required when a valid technique exists for detecting the disease or adverse health effect to which the substance may cause exposure, and the likelihood of that condition arising under the conditions of work is foreseeable. The EU CMRD (Article 14, EU) requires that individual exposure and health surveillance records be maintained for at least 40 years after the last known exposure — a recognition that the latency period for occupational cancer can span most of a working lifetime and well into retirement.
A well-designed health surveillance programme includes four components. First, a baseline health assessment before a worker is assigned to tasks involving carcinogen exposure, establishing their pre-exposure health status. Second, periodic surveillance during employment at intervals appropriate to the carcinogen and exposure level. Third, post-exposure assessment when a worker is removed from carcinogenic tasks. Fourth, post-employment follow-up — the component most frequently missing from employer programmes.
The post-employment gap is a structural weakness. Many employers maintain surveillance records only for the duration of employment, losing critical long-term exposure data when workers change jobs, retire, or are made redundant. In the EU, the 40-year record retention requirement addresses this legally, but compliance depends on employers having systems capable of maintaining records for decades after the employment relationship ends. Exposure registries — such as Finland’s ASA Register and France’s occupational cancer tracking systems — provide a national infrastructure for long-term follow-up that employer-level records alone cannot achieve.
Emerging approaches in biological monitoring include DNA adduct analysis, which detects carcinogen-DNA binding as an early marker of genotoxic damage. These methods remain largely research-stage for most carcinogens and are not yet standard in routine occupational health surveillance. However, they represent a potential shift from detecting cancer after it develops to detecting biological damage before cancer initiates.
Field Test: Ask your occupational health team: what happens to a worker’s exposure records when they leave the company? If the answer involves shredding, archiving in inaccessible storage, or “we don’t know,” the programme has a critical gap. Records for carcinogen-exposed workers should be maintained, accessible, and transferable for decades beyond employment.
What Are the Emerging Issues in Occupational Cancer?
Several developments are reshaping the occupational cancer landscape, and practitioners who are aware of them now will be ahead of regulatory compliance curves rather than reacting to them.
The 6th revision of the EU CMRD represents the most significant recent regulatory expansion. In December 2025, the EU Council agreed its negotiating position adding welding fumes to Annex I of the Directive for the first time, alongside new binding OELs for cobalt compounds, polycyclic aromatic hydrocarbons, 1,4-dioxane, and isoprene. The revision also extends reprotoxic substance definitions. The European Commission’s impact assessment projects that these changes will prevent approximately 1,700 lung cancer cases over a 40-year period (Council of the EU, 2025). For practitioners, the welding fumes inclusion is particularly consequential — welding is performed across virtually every industrial sector, and the regulatory recognition of welding fumes as a carcinogenic exposure will trigger new control, monitoring, and surveillance obligations for millions of workers.
The protection of healthcare workers from hazardous medicinal products is a paradigm-shifting development. Directive 2022/431 (EU) expanded the CMRD to cover HMPs — primarily cytotoxic drugs used in oncology, immunology, and rheumatology that are themselves carcinogenic, mutagenic, or reprotoxic. In early 2025, the European Commission published the definition and indicative list of HMPs containing CMR substances, giving employers a concrete reference for identifying which drugs trigger CMRD obligations. This expansion acknowledges that carcinogen exposure is not limited to “dirty” industrial settings but occurs in clinical environments where the hazard has historically been treated as incidental to patient care.
Climate change and occupational UV exposure is an area of growing concern. Rising UV radiation intensity in many working latitudes increases non-melanoma skin cancer risk for outdoor workers in agriculture, construction, utilities, and transport. The WHO/ILO Joint Estimates have begun quantifying this burden, and several jurisdictions are developing specific guidance on UV exposure management for outdoor workers — a carcinogen that requires fundamentally different control approaches (scheduling, shade structures, protective clothing, sunscreen programmes) from the chemical and particulate carcinogens that dominate traditional industrial hygiene.
Technological advances in exposure assessment are progressing rapidly. Wearable personal exposure monitoring devices capable of real-time measurement of specific carcinogens are moving from research prototypes toward commercial availability. Exposome-based research approaches — which assess the totality of occupational and non-occupational exposures over a lifetime — are challenging the single-agent, single-outcome model that current regulation is built on. While these tools are not yet standard practice, they represent the direction in which occupational cancer prevention is heading.

Frequently Asked Questions
Conclusion
Occupational cancer kills an estimated 799,000 workers every year (ILO/ICOH, 2022), and most of those deaths were preventable at the point of exposure — decades before the diagnosis. That is not a statistical abstraction. It is 799,000 people whose working conditions included carcinogenic exposures that could have been eliminated, substituted, enclosed, or ventilated away, but were not. The latency period does not reduce the moral weight of those failures. It increases it, because it means the people making control decisions today will never see the consequences of their choices in their own careers.
The knowledge, the regulatory frameworks, and the control technologies to prevent the vast majority of occupational cancers already exist. What is missing, consistently, is the operational urgency that chronic hazards fail to generate. Acute hazards demand attention because they punish immediately. Carcinogen exposure permits decades of complacency before presenting its bill. Closing that urgency gap — treating an uncontrolled carcinogenic exposure with the same seriousness as an unguarded moving part — is the single highest-impact change an HSE professional can make.
For practitioners reviewing their own programmes: does your exposure monitoring cover all carcinogenic agents present on your site, not just those with legal PELs? Are your engineering controls maintained, tested, and verified to current performance standards — or were they adequate at installation and assumed adequate since? Do your health surveillance records follow workers beyond employment, meeting the 40-year retention standard that reflects the biological reality of latency? These are not aspirational questions. They are the minimum standard for a programme that treats occupational cancer prevention as what it is — a matter of life and death, deferred in time but not in consequence.
Regulatory content in this article reflects general HSE professional understanding of the cited jurisdictions’ requirements as referenced. It is not legal advice. Specific compliance questions, enforcement situations, or prosecution risk should be directed to qualified legal counsel in the applicable jurisdiction.

