TL;DR
- If a worker’s dermatitis improves on leave and returns at work → the exposure is occupational, and a COSHH or chemical risk assessment review is overdue.
- If the condition responds to dose reduction (shorter contact time, diluted solutions) → irritant contact dermatitis is likely the primary driver, and engineering or substitution controls can make a measurable difference.
- If even trace exposure triggers a flare-up after a symptom-free period → allergic sensitization has occurred, complete avoidance of the specific allergen is the only reliable control, and patch testing through an occupational dermatologist is the diagnostic step.
- If your control program starts and ends with “wear gloves” → you are defaulting to the lowest tier of the hierarchy, and the glove material itself may be causing the problem.
Occupational dermatitis is skin inflammation caused or aggravated by workplace exposures — predominantly affecting the hands — and is the most common occupational skin disease globally. It takes two main forms: irritant contact dermatitis, which accounts for roughly 80% of cases and results from direct chemical damage to the skin barrier, and allergic contact dermatitis, an immune-mediated response triggered after sensitization to a workplace allergen. The distinction between these two forms is not academic — it determines whether reducing exposure dose will work or whether the allergen must be completely eliminated.
What Is Occupational Dermatitis and Why Does It Matter?
Between 6.7% and 10.6% of workers are estimated to have work-related dermatitis (Rydz et al., 2024, Contact Dermatitis), yet only a fraction of those cases ever reach a compensation register or specialist diagnosis. In the UK alone, approximately 876 new cases of work-related contact dermatitis are diagnosed by dermatologists annually, against roughly 16,000 self-reported cases (HSE UK, 2019 data reported 2024). That gap between clinical confirmation and actual prevalence tells you something important about how this disease hides in plain sight across workplaces.
Occupational dermatitis is inflammation of the skin caused or worsened by substances or conditions encountered at work. Over 90% of occupational skin disease is contact dermatitis, and it predominantly affects the hands — the body’s primary point of contact with chemicals, water, and irritants. The burden is not only personal. Contact dermatitis was estimated to cost $1.5 billion annually in the US alone in combined lost wages, reduced productivity, and healthcare spending (Lee et al., 2025, International Journal of Dermatology, citing 2013 data). More critically, studies tracking affected workers over a decade found that up to 50% still had skin problems long after their initial diagnosis.
The pattern that recurs across sectors is depressingly consistent: mild early symptoms — dryness, occasional redness on the knuckles or between the fingers — are dismissed as “just dry skin” or accepted as normal for the job. By the time health surveillance or a GP identifies the condition, chronic damage to the skin barrier has already narrowed the window for effective intervention. That window is front-loaded. Early recognition and early control adjustment determine whether a worker recovers fully or lives with a condition that follows them for years.

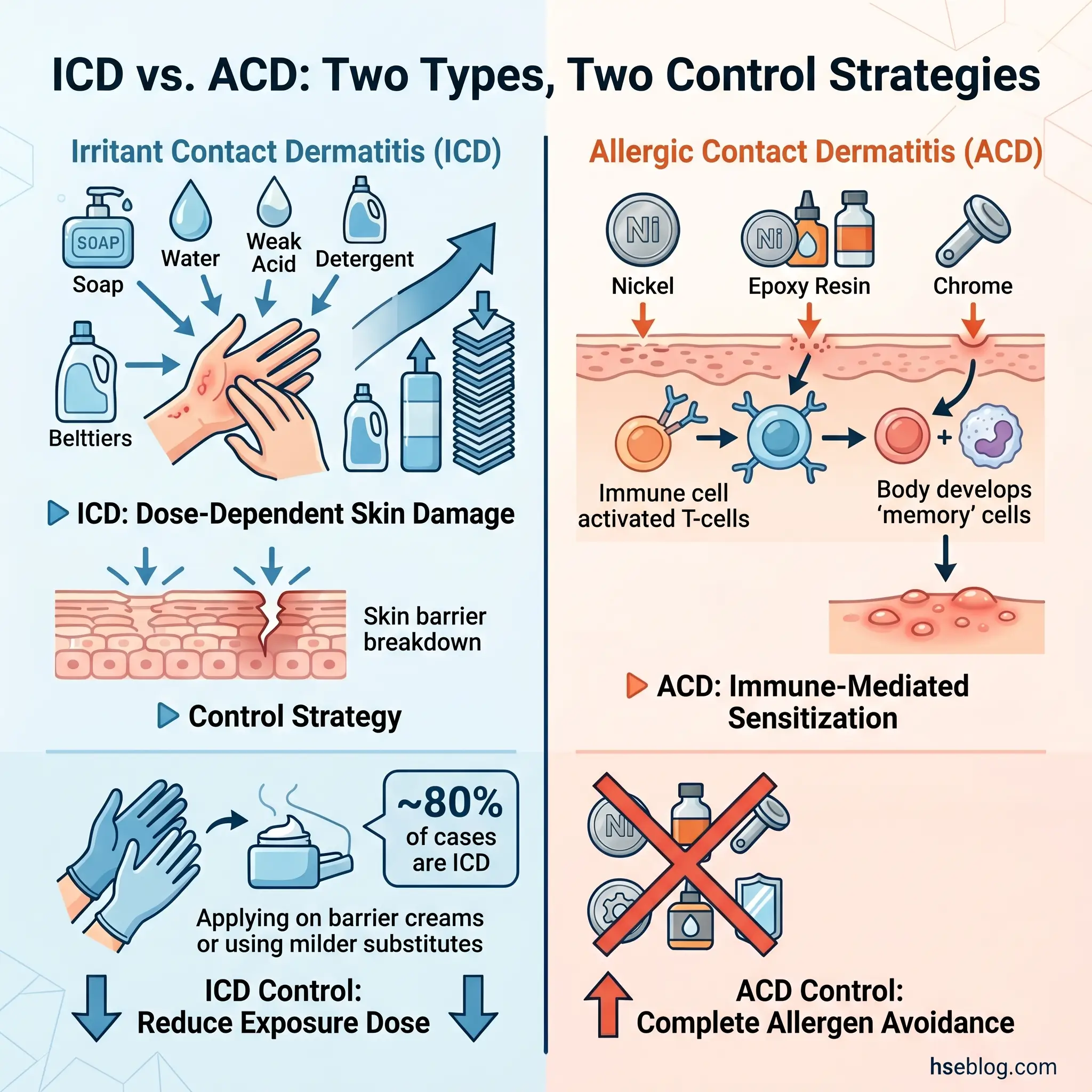
Irritant Contact Dermatitis vs. Allergic Contact Dermatitis: Why the Distinction Changes Your Controls
The single most consequential question in managing occupational dermatitis is not “does this worker have dermatitis?” — it is “which type, and what drives it?” The answer dictates whether your control measures will actually work.
Irritant contact dermatitis (ICD) is a direct toxic injury to the skin. A chemical or physical agent damages the skin barrier through cumulative exposure — the higher the dose, the longer the contact, the more concentrated the substance, the worse the damage. There is no immune system involvement. This is straightforward dose-response toxicology applied to the skin’s outermost layers. Because ICD is dose-dependent, controls that reduce exposure intensity genuinely reduce risk: shorter contact duration, lower concentrations, better rinsing protocols, and effective barrier measures all make a measurable difference.
Allergic contact dermatitis (ACD) operates on an entirely different mechanism. It is a Type IV delayed hypersensitivity reaction — the immune system develops a specific response to an allergen after an initial sensitization phase that can last weeks or months. During that latency, the worker tolerates the substance with no symptoms, creating a false sense of safety. Once sensitized, even trace quantities of the allergen trigger an inflammatory response. Dose reduction is no longer the answer. The only reliable control is complete avoidance of the specific allergen.
This distinction has direct consequences for which tier of the hierarchy of controls you reach for. A control program that treats all occupational dermatitis as irritant — diluting chemicals, reducing contact time, issuing barrier cream — will fail spectacularly when the underlying driver is allergic sensitization. The worker’s condition worsens despite “following the plan,” and the safety professional is left troubleshooting the wrong problem.
The two types frequently coexist. A worker with irritant-damaged skin from wet work has a compromised barrier that permits allergens to penetrate more easily, accelerating sensitization to substances they previously tolerated without issue. Wet work itself carries a 1.56x increased risk of ICD (OR: 1.56, 95% CI: 1.21–2.01) based on meta-analysis of work-related risk factors (systematic review, Contact Dermatitis, 2023).
| Feature | Irritant Contact Dermatitis (ICD) | Allergic Contact Dermatitis (ACD) |
|---|---|---|
| Mechanism | Direct chemical/physical skin damage | Type IV immune hypersensitivity |
| Dose-response | Yes — dose-dependent, cumulative | No — trace amounts trigger reaction once sensitized |
| Onset latency | Hours to days after sufficient exposure | Sensitization phase: weeks to months; elicitation: 24–72 hours |
| Common agents | Water (wet work), soaps, solvents, cutting fluids, acids/alkalis | Epoxy resins, rubber accelerators, nickel, chromium (cement), formaldehyde releasers, PPD |
| Prognosis | Generally good if exposure is reduced/eliminated | Guarded — sensitization is permanent; symptoms managed through avoidance |
| Primary control approach | Dose reduction: shorter contact, dilution, substitution, barrier measures | Complete allergen avoidance: substitution or redeployment |
Watch For: The practitioner trap where a worker’s dermatitis is assumed to be purely irritant — because ICD is statistically more common — and managed with barrier cream and reduced contact time. Meanwhile, the actual driver is allergic sensitization to a rubber accelerator in the worker’s protective gloves. The prescribed PPE is itself the cause. This misdiagnosis pattern is well-documented in occupational dermatology literature and illustrates why patch testing is essential when standard ICD controls fail to resolve symptoms.
How the Damaged Skin Barrier Amplifies Both Types
Compromised skin lowers the threshold for both irritant injury and allergen penetration. Workers with pre-existing atopic dermatitis carry a 2.44x increased risk of developing ICD (OR: 2.44, 95% CI: 1.89–3.15) according to a 2023 systematic review published in Contact Dermatitis. HSE UK guidance identifies wet work exceeding two hours per day — or more than 20 hand washes per shift — as a significant risk factor, a threshold not replicated in OSHA guidance, which lacks a specific wet-work exposure benchmark. The interaction effect compounds the problem: irritant exposure weakens the barrier, which facilitates allergic sensitization to substances the worker previously tolerated without reaction. Pre-employment health questionnaires that identify atopic history are not bureaucratic formalities — they identify workers who need enhanced monitoring from the outset.
High-Risk Industries and Occupations
A flat list of “industries with dermatitis risk” is less useful than understanding the exposure categories that recur across sectors. When conducting a dermatitis risk assessment, the question is not “which industry am I in?” but “which exposure type dominates, and how long does skin contact last?”
Wet-work-dominated exposures drive the highest volume of occupational dermatitis cases. Healthcare workers, food handlers, caterers, cleaning and janitorial staff, and hairdressers share the same fundamental risk: prolonged or repeated skin contact with water, compounded by soaps, detergents, and frequent handwashing. The post-pandemic period amplified this dramatically — the surge in hand hygiene frequency and extended glove-wearing time among healthcare workers caused a well-documented increase in occupational hand dermatitis, prompting new research into workplace intervention effectiveness and gentler hand hygiene substitutes (Ferrari et al., 2025, Diseases; AlEdani et al., 2024, Cureus).
Chemical-exposure-dominated settings introduce different risk profiles. Construction workers handling cement encounter hexavalent chromium — a potent sensitizer. Manufacturing workers in metalworking or automotive repair face cutting fluids, lubricants, and solvents. Printing operations expose workers to inks and solvent-based cleaners. In these environments, the chemical’s specific hazard classification — particularly GHS hazard statements H315 (skin irritation), H317 (skin sensitization), and H314 (serious skin damage) on the Safety Data Sheet — determines whether the primary risk is irritant or allergic.
Mixed-exposure environments — agriculture, laboratory work — combine chemical contact with biological agents, UV radiation, and plant-derived allergens, making the risk assessment more complex.
A pattern consistently overlooked in risk assessments: the highest-risk moment in many operations is not during the primary chemical task but during cleanup. Workers remove PPE, use aggressive skin cleaners or solvent-based hand wipes, and expose already-irritated skin to further chemical insult. A risk assessment that stops at the handling task and ignores the decontamination routine misses a significant exposure pathway.
Recognizing Occupational Dermatitis: Symptoms and Diagnostic Approach
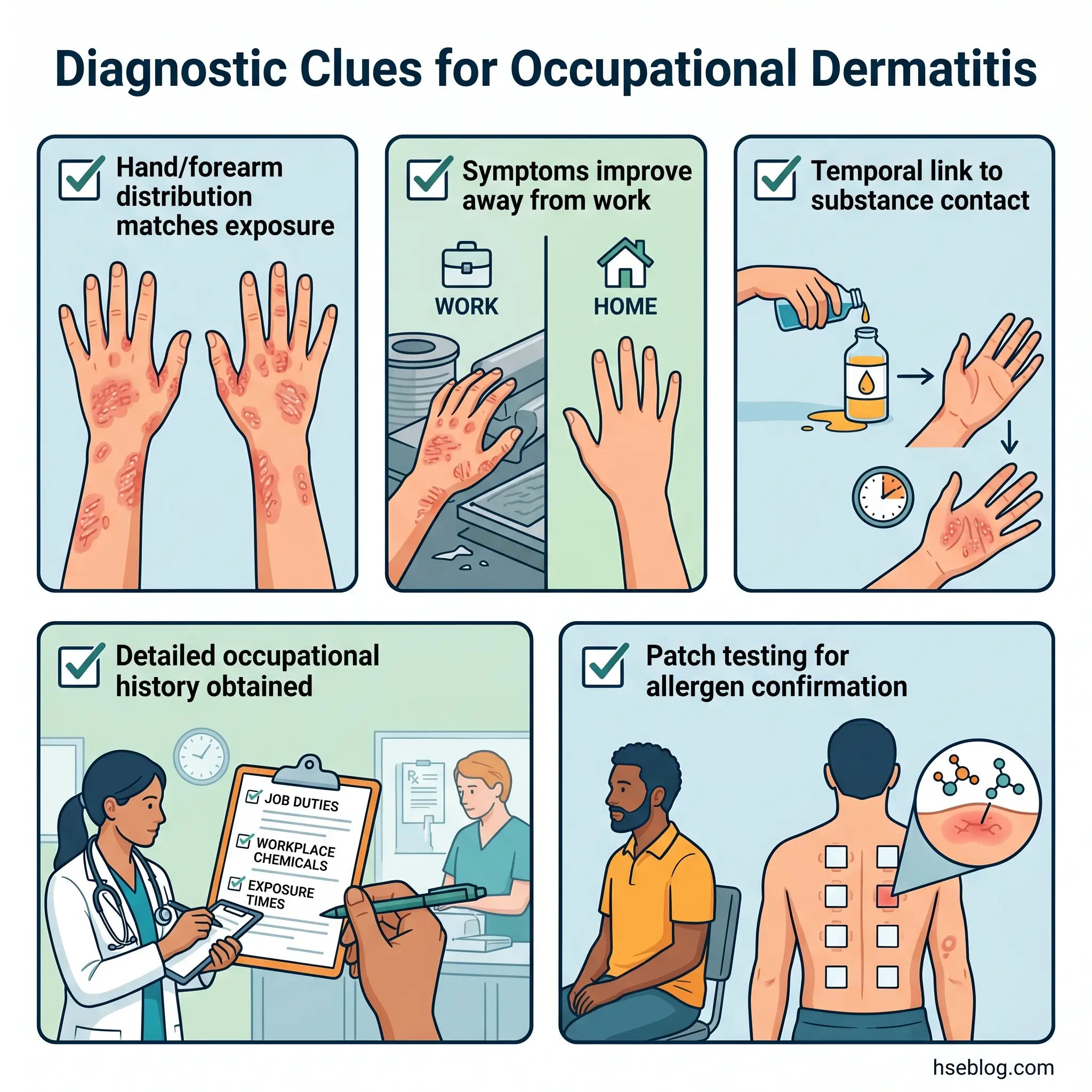
The clinical presentation of occupational contact dermatitis — redness, itching, dryness, cracking, blistering, fissuring — overlaps substantially with non-occupational skin conditions. The diagnostic reasoning that distinguishes occupational origin rests on three pillars: anatomical distribution matching the exposure site, temporal relationship with work, and a detailed occupational history.
Hands are the dominant site. Dorsal surfaces, finger web spaces, and forearms — the areas most frequently in contact with chemicals and water — are the primary distribution pattern. When dermatitis appears on exposed skin in a pattern that maps onto known contact points with a workplace substance, the occupational link strengthens.
The “holiday test” is a practical diagnostic indicator: symptoms that improve during time away from work and return upon resuming duties are strongly suggestive of occupational causation, though not conclusive on their own — non-occupational triggers can coincidentally align with work schedules.
Patch testing is the confirmatory investigation for allergic contact dermatitis. Performed by dermatologists, it involves applying standardized test allergens under occlusive patches for 48 hours, with readings at both 48 and 96 hours to identify delayed hypersensitivity reactions. The critical caveat for safety professionals: patch testing identifies specific ACD allergens but does not diagnose ICD, which remains a clinical and exclusion diagnosis.
Audit Point: In practice, many cases are mixed — a worker exposed simultaneously to wet work (irritant) and rubber glove chemicals (allergen). The diagnostic question for the occupational health practitioner is not a binary “irritant or allergic?” but rather “what is the contribution of each mechanism, and which exposures can be eliminated or substituted?”
Content covering health surveillance, patch testing, and skin symptom assessment is for HSE practitioner reference. It is not medical advice. Workers with specific symptoms or exposure concerns should consult an occupational physician or qualified medical professional.
Workplace Controls: Applying the Hierarchy to Dermatitis Prevention
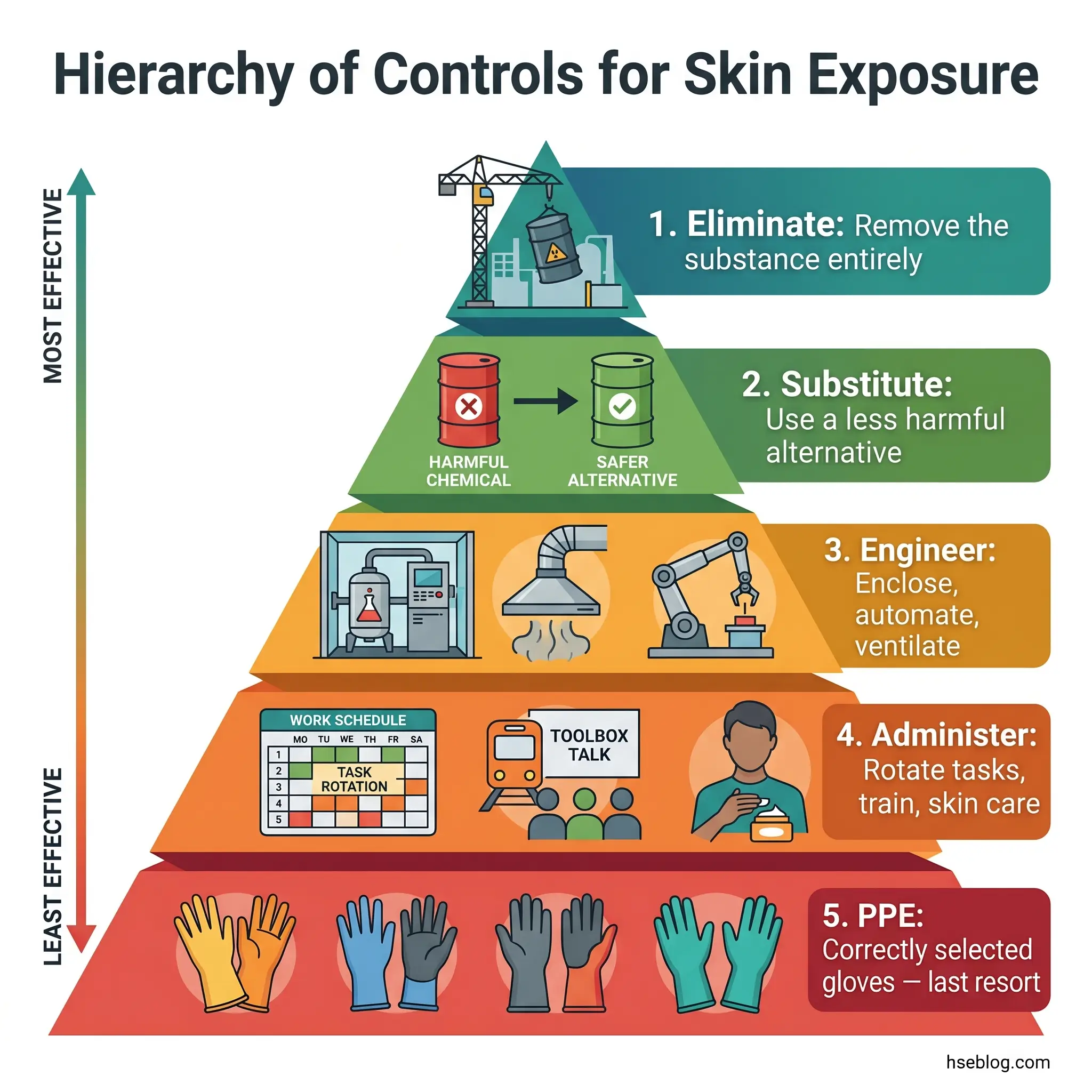
Every regulatory framework covering dermal chemical exposure — COSHH Regulation 7 (UK), the OSHA Hazard Communication Standard (US, 29 CFR 1910.1200), and the EU Chemical Agents Directive 98/24/EC — requires employers to assess dermal exposure risk and apply controls. The hierarchy of controls is not a suggestion; it is the structural framework that determines whether a dermatitis prevention program has legal and operational credibility. The HSE guidance on managing skin exposure risks provides one of the clearest Tier 1 frameworks for this approach.
Elimination removes the substance entirely. Mechanical paint stripping instead of solvent-based methods. Water-jet cutting replacing chemical degreasing. When elimination is feasible, it resolves the dermatitis risk at source — no amount of downstream control matches it.
Substitution replaces a high-risk substance with a lower-risk alternative. Chromium-reduced cement prevents hexavalent chromium sensitization in construction. Formaldehyde-free preservatives in metalworking fluids eliminate a known sensitizer. Switching from harsh detergent-based hand cleansers to gentler, pH-neutral alternatives reduces irritant load. Substitution decisions require checking that the replacement does not introduce a different skin hazard — the Safety Data Sheet for the substitute needs the same scrutiny as the original.
Engineering controls reduce skin contact through physical design: enclosed dispensing systems, automated mixing, splash guards, local exhaust ventilation that prevents airborne chemical deposition on exposed skin, and mechanical parts-washing equipment that replaces manual cleaning in solvent baths.
Administrative controls manage exposure duration and support skin health: job rotation to limit cumulative contact time, scheduled skin recovery breaks, structured skin care programs (see below), and training on correct hand-washing technique — gentle soap, thorough rinsing, patting dry rather than rubbing, immediate moisturizer application.
PPE — the last resort, not the first response. Chemical protective gloves are the most commonly deployed control for occupational dermatitis, and the most commonly misapplied. Gloves issued without reference to the specific chemical, without verification of breakthrough time, and without considering whether the glove material itself is an allergen source do not constitute adequate control. They constitute a liability.
Field Test: Ask a straightforward question about your site’s dermatitis controls: at which tier of the hierarchy does the program actually operate? If the honest answer is “we give them gloves and barrier cream,” the program is anchored at the bottom two tiers — and the three tiers above, where the most effective controls sit, have been skipped.
Glove Selection: Getting It Right
Glove selection for dermatitis prevention requires matching the glove material to the specific chemical exposure — not defaulting to a generic “chemical-resistant” glove. EN ISO 374:2016+A1:2018 classifies chemical protective gloves into Type A, B, and C based on permeation resistance against a set of 18 test chemicals, with breakthrough times that determine how long the glove provides effective protection against a given substance.
The practical process: identify the specific chemicals from the Safety Data Sheet, consult the glove manufacturer’s permeation data for those chemicals, select the material with the appropriate breakthrough time, and establish a change-out interval that accounts for real-world conditions (temperature, physical abrasion, multi-chemical exposure).
Prolonged occlusive glove wearing — particularly nitrile or latex without cotton liner gloves — creates its own irritant dermatitis problem. Trapped moisture and heat macerate the skin, weakening the barrier. This is the “inside the glove” problem, and it is responsible for a significant proportion of hand dermatitis in workers who wear gloves continuously for hours.
Rubber chemicals in gloves — accelerators such as thiurams and carbamates used in the vulcanization process — are themselves among the most common occupational allergens. Latex protein allergy and rubber accelerator allergy are distinct conditions requiring different glove alternatives. A worker with thiuram allergy needs an accelerator-free glove, not simply a “non-latex” glove. The CCOHS guide to allergic contact dermatitis in the workplace provides accessible reference material on common occupational allergens organized by exposure context.
Skin Care Programs: The Overlooked Control Layer
A structured skin care regime — pre-work barrier or conditioning cream, gentle cleansing during work, and after-work emollient or moisturizer — is an administrative control that supports barrier integrity. It does not replace higher-tier controls, and the evidence for barrier creams specifically preventing allergic contact dermatitis is limited. Where they contribute is in reducing the severity and frequency of irritant dermatitis by maintaining the skin’s natural protective function.
Emollients should be applied after every hand wash, not reserved for the end of the shift. Abrasive hand cleaners and solvent-based hand wipes — ironically common in industries with the highest dermatitis rates — are themselves significant irritants. Replacing them with gentler alternatives is a substitution decision that costs very little and reduces irritant load substantially. The operational key: skin care works when it is built into the work routine with accessible products at the point of use, not when it is treated as a personal responsibility and a tube of cream sits forgotten in a locker.
Health Surveillance for Occupational Dermatitis
Under COSHH Regulation 11 (UK), health surveillance is legally required when workers are exposed to substances capable of causing identifiable disease — including dermatitis — and adequate control cannot be guaranteed by other means alone. This is a secondary prevention measure: it does not replace exposure controls, but it detects early cases before chronic, irreversible skin damage sets in.
The surveillance framework described in HSE health surveillance guidance for occupational dermatitis (G403) includes a pre-employment baseline questionnaire to identify atopic history and existing skin conditions, regular skin inspections by a trained “responsible person,” defined escalation criteria triggering referral to an occupational health professional or dermatologist, and systematic record-keeping that enables trend analysis.
The responsible person conducting routine skin checks does not need to be a clinician. They need to be trained to recognize early signs — dryness, redness, cracking on the hands and forearms — and to follow the escalation pathway when those signs appear. G403 recommends baseline assessment within six weeks of starting a relevant role, with ongoing check frequency guided by the occupational health professional’s risk assessment.
Where health surveillance consistently fails is at the feedback loop. Skin checks are conducted, records are kept, early cases are identified — and then the response is directed solely at the affected individual: “use more moisturizer,” “see your GP.” The finding that three workers out of twelve in the same area show early-stage hand dermatitis is not a personal health issue for three people. It is a control-failure signal for the entire operation. That finding should trigger an immediate reassessment of the chemical risk assessment, exposure controls, and work practices for every worker in the area — not just a referral for the symptomatic three.
The Fix That Works: Build a standing protocol: any new dermatitis case identified through surveillance triggers a control review for the task, not just treatment for the worker. Document the link between surveillance findings and control actions. Auditors under COSHH look for exactly this feedback mechanism — evidence that health surveillance data drives operational change.
Regulatory Framework Across Jurisdictions
Regulatory content here reflects general HSE professional understanding of multi-jurisdictional requirements as of 2025. It is not legal advice. Specific compliance questions, enforcement situations, or prosecution risk should be directed to qualified legal counsel in the applicable jurisdiction.
Employers operating across borders face regulatory frameworks that share the same objective — preventing occupational skin disease — but differ substantially in structure and specificity.
United Kingdom: COSHH Regulations 2002 (as amended) place the duty on the employer to prevent or adequately control exposure to substances hazardous to health, with “adequate control” defined through the hierarchy (Regulation 7). Health surveillance under Regulation 11 is mandatory when dermatitis risk exists and cannot be fully controlled. Supporting guidance in HSG262 covers identifying and managing occupational skin exposure. The UK approach is “goal-setting” — the employer must demonstrate adequate control rather than meeting a prescriptive checklist.
United States: OSHA does not have a single dermatitis-specific standard. Instead, obligations flow through the Hazard Communication Standard (29 CFR 1910.1200), which requires communication of skin hazards via GHS-aligned labelling and Safety Data Sheets, plus worker training. Substance-specific standards apply to certain skin-designated chemicals — formaldehyde (1910.1048) and hexavalent chromium (1910.1026) among them. The General Duty Clause provides a catch-all obligation. The 2025 review of OSHA-regulated substances causing occupational dermatitis (Lee et al., International Journal of Dermatology) systematically maps OSHA-regulated toxic substances to dermatitis mechanisms and exposure case data from 2017–2023, providing the most current synthesis available.
European Union: The Chemical Agents Directive 98/24/EC requires employers to assess dermal exposure risk from hazardous chemical agents and apply the hierarchy of controls. The CLP Regulation classifies substances as skin irritants (H315), serious skin damage (H314), or skin sensitizers (H317), directly informing the employer’s risk assessment. EU-OSHA provides sector-specific guidance on skin disease prevention, including wet-work thresholds in healthcare and cleaning sectors.
Canada: WHMIS aligns with GHS for hazard communication. CCOHS publishes practical guidance on occupational contact dermatitis prevention and allergen identification by occupation.
Australia: Safe Work Australia’s model codes of practice cover chemical risk management with specific guidance on wet-work dermatitis prevention.
The practical consequence for multinational employers: where thresholds differ, the stricter standard should govern. The UK’s wet-work threshold (more than two hours per day or more than 20 hand washes) is more specific than anything in OSHA’s framework — it provides a concrete, defensible benchmark regardless of operating jurisdiction.

What Happens When Controls Fail: Prognosis and Return-to-Work Considerations
Irritant contact dermatitis generally carries a better prognosis — when the irritant exposure is reduced or eliminated, the skin barrier can recover, sometimes fully. The timeline depends on severity and duration of prior exposure, but for mild-to-moderate ICD caught early, meaningful improvement within weeks of effective exposure control is common.
Allergic contact dermatitis presents a harder reality. Sensitization is permanent. The immune system does not “forget” the allergen. For certain substances — hexavalent chromium and epoxy resins among the most challenging — dermatitis can persist for years even after complete avoidance. Published follow-up studies consistently show that a significant proportion of workers with confirmed ACD continue to experience symptoms a decade after diagnosis, particularly when sensitization occurred to ubiquitous allergens that are difficult to avoid entirely.
Return-to-work decisions require occupational health assessment — not a line manager’s judgment call. The core question is whether the specific allergen or irritant can be completely eliminated from the worker’s role. If substitution or engineering controls can remove the exposure, return to the original role may be feasible. If they cannot — and this is frequently the case with potent sensitizers in roles where the substance is fundamental to the process — redeployment to a role without the specific exposure becomes a reasonable adjustment under disability and equality legislation in many jurisdictions.
The tension between a worker’s desire to return to their original role and the medical reality that re-exposure will cause relapse requires honest communication. There is no single correct answer — the decision depends on whether the exposure can be truly eliminated from that specific job, not merely reduced. Workers’ compensation and reporting obligations add another layer: RIDDOR reporting applies in the UK for confirmed occupational dermatitis cases; in the US, recording on the OSHA 300 log is required for work-related skin disorders meeting the recording criteria.
Frequently Asked Questions
Conclusion
What the industry consistently gets wrong with occupational dermatitis is treating it as a minor nuisance rather than a condition with a narrow intervention window and, for allergic cases, permanent consequences. The single highest-impact change a safety professional can make is to stop treating all occupational dermatitis as irritant by default. The clinical distinction between irritant and allergic contact dermatitis is not a dermatologist’s concern alone — it determines whether your control program will succeed. A dose-reduction strategy applied to an allergic sensitization problem will fail every time, and the worker’s condition will worsen under your watch.
The second failure is structural. Health surveillance that identifies cases without feeding findings back into control reassessment is a record-keeping exercise, not prevention. Glove programs that issue generic “chemical-resistant” gloves without matching the material to the specific chemical — and without considering that the glove itself may be the allergen source — are control programs in name only. These are not knowledge gaps; they are implementation failures that persist because occupational dermatitis lacks the dramatic immediacy of a fall or an explosion.
Occupational skin disease is the most common occupational health condition globally, and the most quietly tolerated. The controls exist, the regulatory frameworks are clear across jurisdictions, and the diagnostic tools are well-established. What remains is the operational discipline to apply them with the same seriousness given to hazards that produce visible, immediate harm. A worker’s hands are their livelihood. Treating their skin with the same engineering rigor as any other workplace hazard is not best practice — it is the minimum standard.