The spirometer sitting on the testing bench looks deceptively simple. A mouthpiece, a sensor, a calibration syringe beside it, a screen displaying two curves — flow-volume and volume-time — waiting for a forced breath. But what that device captures in a six-second blast of exhalation can determine whether a mineral processing operator keeps working at the crusher or gets pulled from the exposure zone. The FEV1 number on that screen is the earliest measurable boundary between reversible lung function change and permanent occupational lung disease. Every reading carries weight because every reading either confirms a worker’s respiratory baseline is holding or signals that something in the air is already causing damage.
The scale of the problem justifies the vigilance. More than 10% of all lung diseases are estimated to result from workplace exposures. In the UK alone, an estimated 11,000 lung disease deaths occur annually linked to past occupational exposure — dominated by COPD, non-asbestos lung cancer, asbestos-related cancer, and mesothelioma. Approximately 2.3 million workers in the United States are exposed to silica at work, and occupational asthma accounts for 15–23% of new adult asthma cases.
These are not abstract numbers. They represent workers whose lung function declined year over year while they were still reporting for shift, still passing through the gate, still breathing the same air. This article maps every regulatory trigger for workplace spirometry testing, walks through the procedural standards that separate valid data from noise, explains how to interpret results with enough depth to catch decline before it becomes disease, and addresses the program-level systems that keep occupational spirometry programs legally defensible and clinically meaningful.

What Is Spirometry and Why Does It Matter in the Workplace?
Spirometry is the most frequently performed pulmonary function test in occupational health, and for good reason — it is the only standardized, reproducible, and cost-effective method for detecting airflow limitation before a worker develops symptoms. The test measures three primary values. FVC (Forced Vital Capacity) is the total volume of air a person can forcefully exhale after a maximum inhalation — it reflects overall lung size and the ability to fully inflate and deflate the lungs. FEV1 (Forced Expiratory Volume in one second) is the volume expelled in the first second of that forced exhalation — it captures how quickly air moves through the airways and is the most sensitive indicator of early obstruction. The FEV1/FVC ratio compares the two: a declining ratio signals obstructive disease, while a proportionally reduced FVC with a preserved ratio suggests restrictive pathology.
| Parameter | What It Measures | What a Low Value May Indicate |
|---|---|---|
| FVC | Total forced exhaled volume | Restrictive lung disease, fibrosis, chest wall restriction |
| FEV1 | Volume exhaled in first second | Obstructive airway disease, early silicosis, asthma |
| FEV1/FVC | Proportion of lung emptied in one second | Airflow obstruction (COPD, occupational asthma) |
The distinction between screening spirometry and diagnostic pulmonary function testing matters for employers. Workplace spirometry is a screening tool — it identifies workers whose lung function has deviated from their baseline or from expected norms, flagging them for further clinical evaluation. It does not diagnose disease. Full diagnostic PFTs may include lung volumes, diffusing capacity (DLCO), bronchial provocation testing, and imaging. Employers are responsible for the screening; the diagnostic workup belongs to the occupational physician or pulmonologist.
Workplace spirometry serves three tiers of prevention simultaneously. Primary prevention uses baseline spirometry before exposure assignment to document a worker’s starting lung function — this becomes the reference point everything else is measured against. Secondary prevention uses periodic testing to catch early decline while it is still subclinical and potentially reversible through exposure reduction. Tertiary prevention uses spirometry to monitor workers with diagnosed occupational lung disease, guiding fitness-for-duty decisions and medical removal timing.
The exposures that make spirometry testing necessary span nearly every heavy industry: mineral dusts like silica, asite, and coal; metal fumes from welding, smelting, and cadmium processing; organic dusts including cotton, wood, and grain; chemical vapours such as formaldehyde, benzene, and isocyanates; and respiratory sensitisers like flour dust, latex, and enzyme-containing products. Workplace exposures contribute substantially to the burden of multiple chronic respiratory diseases — asthma (16%), COPD (14%), chronic bronchitis (13%), idiopathic pulmonary fibrosis (26%), and hypersensitivity pneumonitis (19%). Those percentages represent preventable fractions. Spirometry is the tripwire that catches the damage early enough to act.
Which Regulatory Standards Require Workplace Spirometry?
No single regulation governs all workplace spirometry testing. Instead, the obligation emerges from a patchwork of substance-specific standards, respiratory protection requirements, and jurisdiction-specific health surveillance frameworks. Understanding which regulation applies to your operation is the first compliance step — and the one most frequently bungled. I have audited facilities running spirometry programs under the wrong standard’s interval, using technicians who met one standard’s training requirement but not another’s. Getting this map right matters.
OSHA Substance-Specific Standards
OSHA mandates spirometry through individual substance standards, each specifying its own triggers, test parameters, and intervals. Here are the critical ones:
- 29 CFR 1910.1053 / 1926.1153 (Respirable Crystalline Silica): Requires spirometry including FVC, FEV1, and FEV1/FVC ratio for workers exposed at or above the action level of 25 µg/m³ for 30 or more days per year. Initial exam within 30 days of assignment. Periodic exams every three years, or more frequently if the PLHCP recommends. Technicians must hold a current certificate from a NIOSH-approved spirometry training course.
- 29 CFR 1910.1001 (Asbestos — General Industry): PFTs including FVC and FEV1 are required for workers exposed above the PEL (0.1 f/cc) or the excursion limit (1.0 f/cc averaged over 30 minutes). Annual testing. Technicians must have completed spirometry training.
- 29 CFR 1910.1043 (Cotton Dust): The original and most technically detailed OSHA spirometry standard. Specifies equipment accuracy (historically ±3%, now updated), maneuver requirements (3–8 forced expirations), and approximately 16 hours of technician training. The July 15, 2019 amendment mandated real-time flow-volume and volume-time curve display and a 0–8 liter BTPS volume range for instruments purchased after May 14, 2020.
- 29 CFR 1910.1027 (Cadmium): Requires PFTs including FVC and FEV1 as part of medical surveillance for exposed workers. Initial and annual testing.
- 29 CFR 1910.1029 (Coke Oven Emissions): PFTs including FVC, FEV1, and FEV1/FVC ratio required for workers exposed above the PEL.
- 29 CFR 1910.1048 (Formaldehyde): PFTs may be required when respiratory protection is used or when the PLHCP determines necessity.
- 29 CFR 1910.1028 (Benzene): PFTs may be triggered as part of medical surveillance for workers exposed above the action level.
- 29 CFR 1910.1024 (Beryllium): Medical surveillance including spirometry for workers with airborne exposure above the action level.
The respiratory protection standard, 29 CFR 1910.134, requires medical evaluation for all respirator users, but spirometry is not automatically required for every worker who wears a respirator. The PLHCP uses a medical questionnaire to determine if spirometry is needed — but substance-specific standards override this, mandating spirometry regardless of the questionnaire outcome.
| Standard | Substance | Spirometry Trigger | Testing Frequency | Technician Training |
|---|---|---|---|---|
| 1910.1053 | Silica | ≥ action level 30+ days/yr | Every 3 years (or per PLHCP) | NIOSH-approved course required |
| 1910.1001 | Asbestos | Above PEL or excursion limit | Annual | Spirometry training required |
| 1910.1043 | Cotton dust | Any exposure requiring controls | Baseline + periodic | ~16-hour training required |
| 1910.1027 | Cadmium | Above action level | Annual | Training required |
| 1910.1029 | Coke oven emissions | Above PEL | Annual | Training required |
| 1910.134 | Any (respirator use) | PLHCP determination | Per PLHCP | Recommended |

COSHH and UK Health Surveillance Requirements
Under the COSHH Regulations 2002, the obligation to perform spirometry does not flow from a substance list in the way OSHA’s framework operates. Instead, where a risk assessment identifies that workers are exposed to respiratory sensitisers or substances known to cause occupational lung disease and exposure cannot be adequately controlled, health surveillance — including spirometry — becomes a legal requirement.
HSE guidance recommends a structured surveillance schedule for workers exposed to respiratory sensitisers: baseline spirometry within four weeks of first exposure, then follow-up at six weeks, three months, six months, and annually thereafter. This front-loaded schedule reflects the rapid onset window for sensitisation-related conditions like occupational asthma.
A colleague running a health surveillance programme at a coatings facility once described the COSHH approach as “the risk assessment does the work that OSHA’s substance list does — but it also means you can’t hide behind the absence of a named standard.” That captures the distinction precisely. If isocyanates are present and workers are exposed, spirometry is required regardless of whether a specific regulation names the concentration threshold.
COSHH health surveillance records must be retained for at least 40 years — a decade longer than the OSHA requirement — reflecting the latency period of many occupational lung diseases.
The 2024 MSHA Silica Rule
The most significant recent development in occupational spirometry regulation is MSHA’s Final Rule on Respirable Crystalline Silica, effective June 17, 2024. The rule requires medical surveillance — including spirometry — for miners exposed above the action level of 25 µg/m³. Compliance dates are phased: metal and non-metal (MNM) mine operators face an April 8, 2026 compliance deadline for medical surveillance provisions, while coal mine operators’ enforcement has been paused pending federal litigation as of early 2025.
This rule closes a regulatory gap that left miners with fewer spirometry protections than their general-industry counterparts. For occupational health programmes serving the mining sector, the 2026 compliance deadline means spirometry infrastructure — equipment, trained technicians, PLHCP agreements — must be in place now. MSHA requires medical records to be preserved for the duration of employment plus six months, a shorter retention period than OSHA’s 30-year standard.
Who Is Qualified to Perform Workplace Spirometry?
Technician qualification is the compliance question that generates the most confusion — and the most audit findings. The short answer is that any person administering spirometry under OSHA’s silica or cotton dust standards must hold a current certificate from a NIOSH-approved spirometry training course. The longer answer involves several critical distinctions.
The NIOSH-approved course is approximately 16 hours of instruction covering respiratory physiology, spirometer instrumentation, test performance technique, quality assessment, and basic interpretation. Passing the course demonstrates baseline competency. But a certificate alone does not ensure ongoing competence. The ATS/ERS 2019 standardization recommends refresher training every three to five years; ACOEM recommends every five years. Neither is legally mandated under current OSHA standards, but a technician producing spirometry data they were trained on a decade ago, on equipment that has since been replaced, using criteria that have been updated, is a program liability.
Audit Point: OSHA’s 2018 interpretation letter confirmed that all personnel administering PFTs under the silica standards — including physicians — must hold a current NIOSH-approved spirometry course certificate. A physician’s medical degree does not exempt them from this requirement.
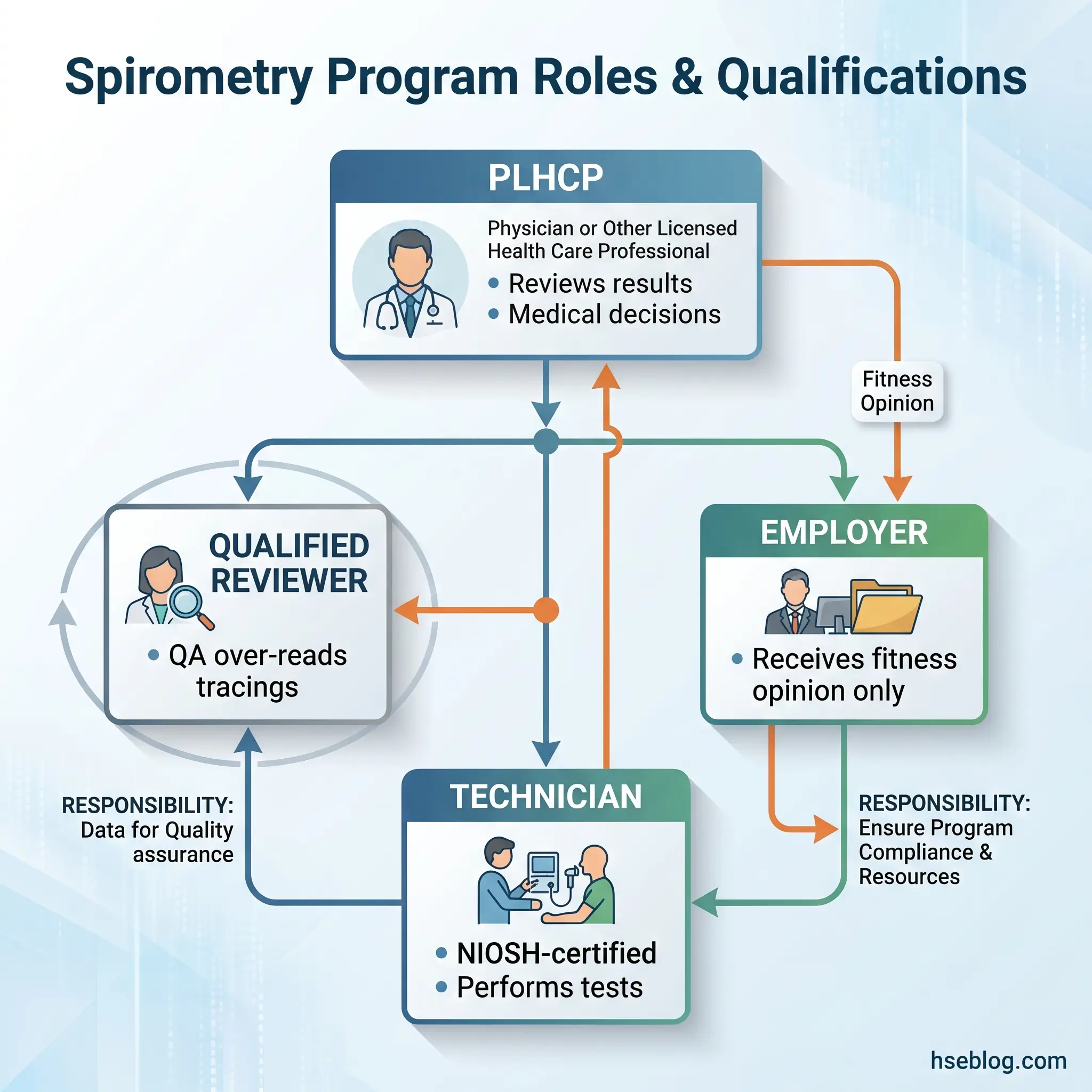
The PLHCP (Physician or Licensed Health Care Professional) oversees the spirometry program but does not necessarily perform the tests. The PLHCP reviews results, makes medical determinations about fitness for duty, and decides whether additional clinical evaluation is warranted. The employer receives only a written medical opinion — not the worker’s clinical data. That confidentiality boundary is regulatory, not optional.
During an equipment vendor demonstration at a mineral processing facility, the vendor’s technician produced textbook-quality curves. A week later, the site’s own technician — trained but out of practice — generated data that a qualified reviewer flagged as uninterpretable in three out of five sessions. Certification is the entry ticket. Competency maintenance is the actual requirement.
Equipment Standards and Calibration Requirements
The 2019 ATS/ERS standardization update tightened the spirometer accuracy requirement from ±3% to ±2.5%, with a corresponding ±0.5% tolerance for the calibration syringe itself. All spirometers used in occupational health programmes must now comply with ISO 26782:2009, the international standard defining validation waveforms and technical performance criteria. If your spirometer predates this standard and has not been verified against the updated requirements, the data it produces may not withstand regulatory scrutiny.
Calibration verification — not calibration itself — must be performed daily before testing begins. The distinction matters. Calibration is the manufacturer’s process of setting the instrument’s measurement accuracy. Calibration verification is the user’s daily confirmation that the instrument is still reading accurately, performed with a 3-liter calibration syringe. The syringe is discharged through the spirometer at low, medium, and high flow rates. If the measured volume deviates from the syringe’s known 3-liter volume by more than the accuracy tolerance, the instrument requires service before any worker testing proceeds.
The following steps outline daily calibration verification:
- Power on the spirometer and allow it to stabilize per the manufacturer’s warm-up specification.
- Attach the 3-liter calibration syringe to the spirometer via the same mouthpiece and filter setup used for worker testing.
- Discharge the syringe at a slow, steady rate. Record the measured volume.
- Repeat at a moderate rate and again at a rapid rate.
- Verify all three readings fall within ±2.5% of 3.0 liters (acceptable range: 2.925–3.075 liters).
- Document the date, time, syringe serial number, measured volumes, ambient temperature, and the technician’s initials in the calibration verification log.
- If any reading falls outside tolerance, remove the spirometer from service and initiate troubleshooting or manufacturer service.
The 2019 Cotton Dust Standard amendment added a requirement that spirometers purchased after May 14, 2020 must display flow-volume and volume-time curves in real time during testing. This is not a convenience feature — real-time curve display allows the technician to identify poor-quality efforts immediately, coaching the worker to correct technique before exhausting the eight-attempt maximum.
Field Test: Pull your most recent calibration verification log. Is every field completed? Is the syringe serial number recorded? Is there a corrective action entry for any out-of-tolerance reading? If any of those answers is no, your spirometry data has a defensibility gap.
Infection control adds another equipment layer. Disposable single-use mouthpieces are mandatory. Bacterial and viral filters should be used between the mouthpiece and the spirometer sensor. Between workers, the mouthpiece and filter assembly is discarded, and external surfaces are cleaned per manufacturer protocol. The spirometer’s internal tubing and sensor should never be exposed to exhaled aerosol when filters are properly placed.
How to Conduct Workplace Spirometry: Step-by-Step Procedure
The best spirometer in the world produces garbage data without proper technique. And technique in occupational spirometry is not just the worker’s technique — it is the technician’s ability to coach, assess, and adapt to each individual’s effort. The OSHA Best Practices guide (OSHA 3637) identifies technician-worker interaction as the single largest variable in test quality. A quiet, encouraging technician who explains, demonstrates, and coaches will consistently produce higher-quality data than a technician who says “blow” and hits record.
Pre-Test Preparation
Before the mouthpiece goes in, several pre-test conditions must be confirmed:
- No smoking or e-cigarette use for at least one hour before testing.
- No vigorous exercise within one hour — elevated respiratory rate and bronchospasm risk compromise baseline readings.
- No heavy meal within two hours — abdominal distension restricts diaphragmatic excursion.
- Loose clothing — belts, tight coveralls, and chest straps restrict thoracic expansion. I have asked workers to loosen their coverall zippers more times than I can count.
- Medication disclosure — particularly bronchodilator use. If a worker used a rescue inhaler that morning, it must be documented because post-bronchodilator spirometry measures a different physiological state than baseline.
The 2019 ATS/ERS update expanded the list of relative contraindications for spirometry: recent thoracic, abdominal, or eye surgery; cardiovascular instability; known pneumothorax; haemoptysis of unknown origin; and recent respiratory infection. These do not automatically prohibit testing but require clinical judgement — typically the PLHCP’s determination.
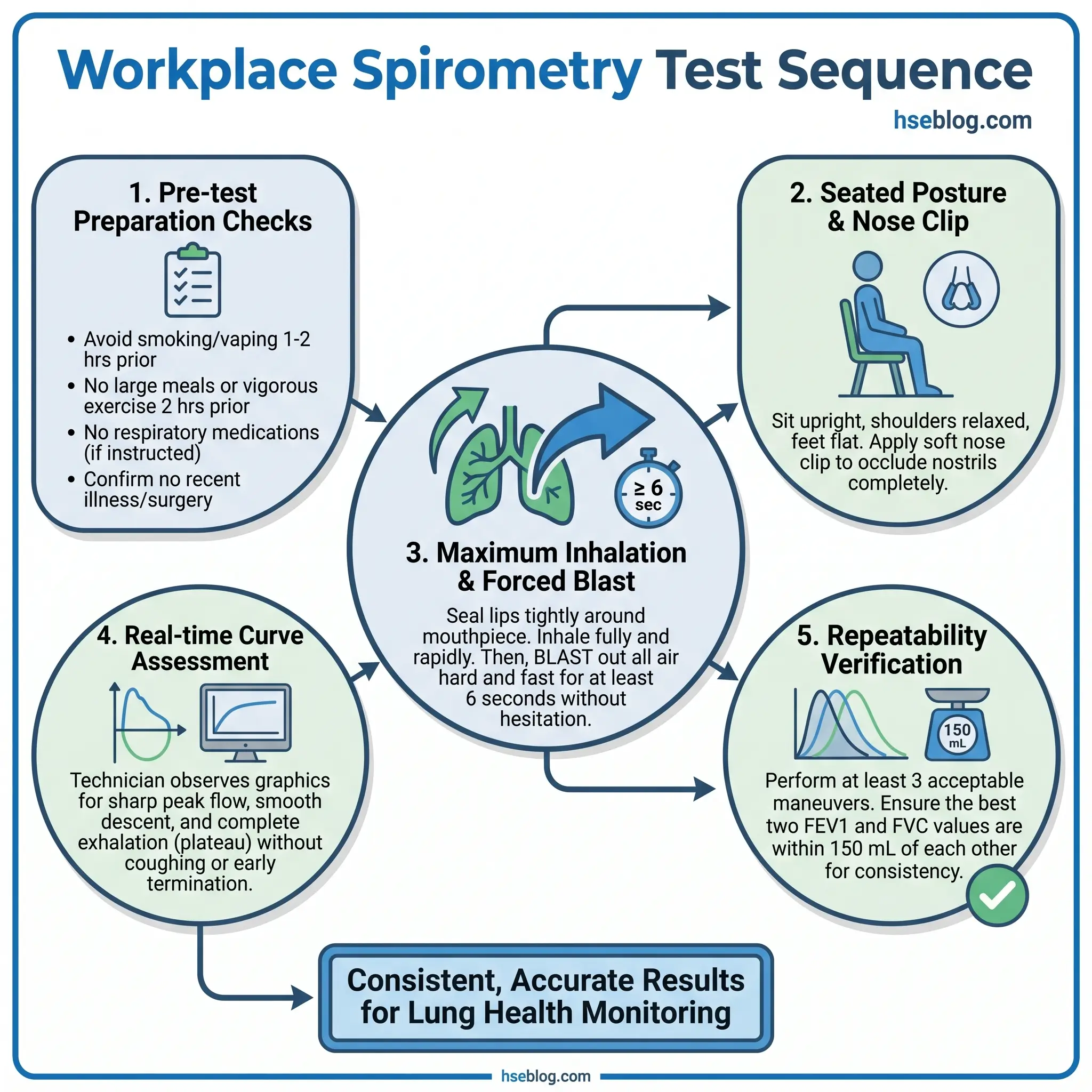
Performing the Maneuver
- Seat the worker upright with feet flat on the floor. Chin slightly elevated, neck slightly extended. Document the posture used — standing versus seated can affect results by up to 7%.
- Apply a nose clip. Demonstrate the maneuver yourself or show a coaching video.
- Instruct the worker to inhale maximally — “Fill your lungs completely, as much air as you possibly can.”
- With mouthpiece sealed tightly around the lips, instruct: “Blast the air out as hard, as fast, and as long as you possibly can — keep going, keep going, keep pushing.”
- Monitor the real-time curves. The flow-volume curve should show a sharp, steep peak flow followed by a smooth descent. The volume-time curve should show a rapid rise followed by a plateau.
- A minimum of three acceptable maneuvers is required. A maximum of eight is permitted per session. More than eight forced expirations risks fatigue-related decline in performance.
- Between maneuvers, allow the worker to rest and breathe normally. Watch for dizziness or light-headedness — syncope during spirometry is rare but real.
Acceptability and Repeatability
Each individual maneuver must meet acceptability criteria: a sharp peak flow (no hesitation start), no cough during the first second, no early termination (exhalation must continue for at least six seconds in adults, or until a clear volume-time plateau is reached), and no air leak at the mouth.
Repeatability is assessed across the session: the two largest FVC values and the two largest FEV1 values should be within 150 mL of each other. The 2019 ATS/ERS update introduced a quality grading system — Grades A through F and U — based on the number of acceptable maneuvers and the degree of repeatability achieved. Grade A represents three or more acceptable maneuvers with repeatability within 150 mL. Grade F indicates no acceptable maneuvers. Grade U means the results are usable but with noted limitations.
Watch For: A worker who achieves a sharp peak flow but consistently terminates exhalation at three to four seconds. This produces an artificially low FVC while the FEV1 may appear normal — mimicking an obstructive pattern that does not exist. Coaching patience into the exhalation is the technician’s job, and it is the single most common reason for uninterpretable data in the field.
Interpreting Workplace Spirometry Results
Interpretation is where occupational spirometry diverges most sharply from clinical practice — and where most workplace programmes fall short. A clinical pulmonologist interprets a single visit against reference norms and clinical context. An occupational health programme must interpret each test both against reference norms and against that worker’s own prior data, across years or decades of employment.
Cross-Sectional Evaluation: Comparing to Reference Values
A single spirometry session is compared to predicted values derived from population reference equations. In the United States, the NHANES III reference set remains the required or recommended reference for occupational testing. For Asian-American workers, a 0.88 scaling factor is applied to predicted FVC and FEV1 values. The ATS 2014 technical standard for spirometry in the occupational setting addressed the use of reference values for diverse populations — getting the correct reference equation matters because an incorrect equation shifts the boundary between “normal” and “abnormal.”
Abnormality is defined by the Lower Limit of Normal (LLN), set at the 5th percentile of the reference population. A worker whose FEV1 falls below the LLN has a result outside the range expected for a healthy individual of the same age, sex, height, and ethnicity. A reduced FEV1/FVC ratio below the LLN indicates an obstructive pattern — suggesting airflow limitation from conditions like occupational asthma, COPD, or early silicosis. A reduced FVC with a preserved ratio suggests a restrictive pattern — potentially indicating pulmonary fibrosis, asbestosis, or chest wall restriction.
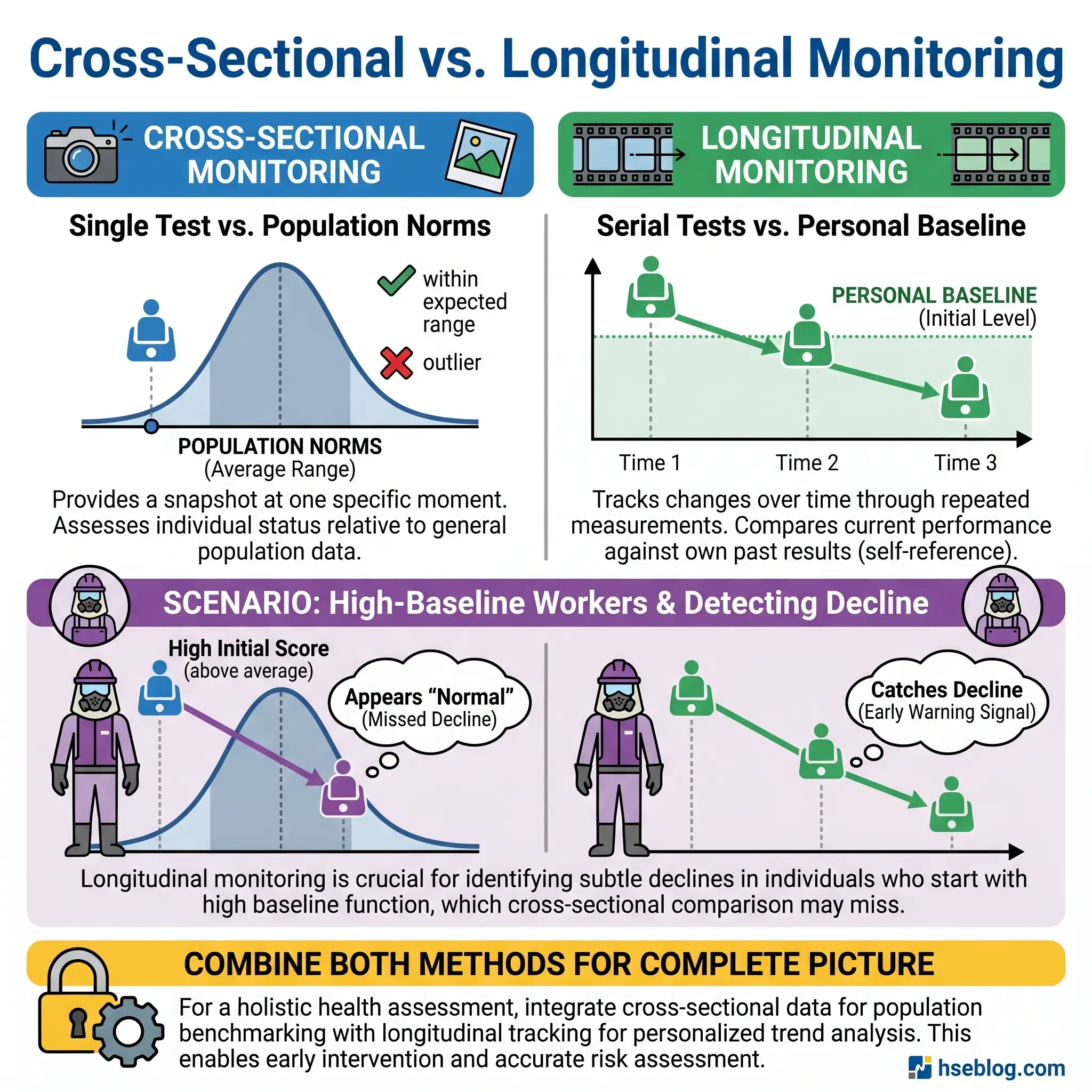
Longitudinal Monitoring: Tracking Changes Over Time
Cross-sectional comparison has a critical blind spot. A worker who starts employment with an FEV1 at 120% of predicted can lose 25% of their lung function and still test “within normal limits.” That worker has experienced a massive, clinically significant decline that a single-test comparison to population norms would miss entirely.
Longitudinal monitoring — tracking each worker’s FEV1 against their own baseline over time — is the more sensitive approach. The baseline spirometry performed before or at the start of exposure becomes the individual’s reference. Subsequent tests are compared to that personal baseline, and the rate of FEV1 decline is calculated.
Normal age-related FEV1 decline is approximately 25–30 mL per year in non-smoking adults. Excessive decline — generally defined as greater than 60 mL per year sustained over several years, or a drop exceeding 15% from baseline — warrants investigation. Some programmes use year-to-year comparison limits; others apply regression-based analysis across five or more data points. The OSHA Best Practices guide recommends that any worker showing a decline of 15% or more in FEV1 from baseline be referred for further evaluation, even if the current value remains above the LLN.
Testing frequency should match the exposure risk profile. For chronic diseases with gradual onset — silicosis, asbestosis — testing every two to three years is generally sufficient. For rapidly developing conditions like occupational asthma, flavouring-related bronchiolitis obliterans, or sensitiser-induced disease, testing every six to twelve months is appropriate. The COSHH front-loaded schedule for sensitiser exposure reflects this urgency.
“If the number is still in the green zone, why are we worried?” a shift supervisor once asked after reviewing an operator’s spirometry trend. The answer was on the chart: a steady, consistent decline of 80 mL per year over four years. Still above the LLN. Still asymptomatic. But on a trajectory toward permanent obstruction within another three to four years if the exposure continued. That conversation changed the ventilation investment on that processing line.
Quality Assurance in Occupational Spirometry Programs
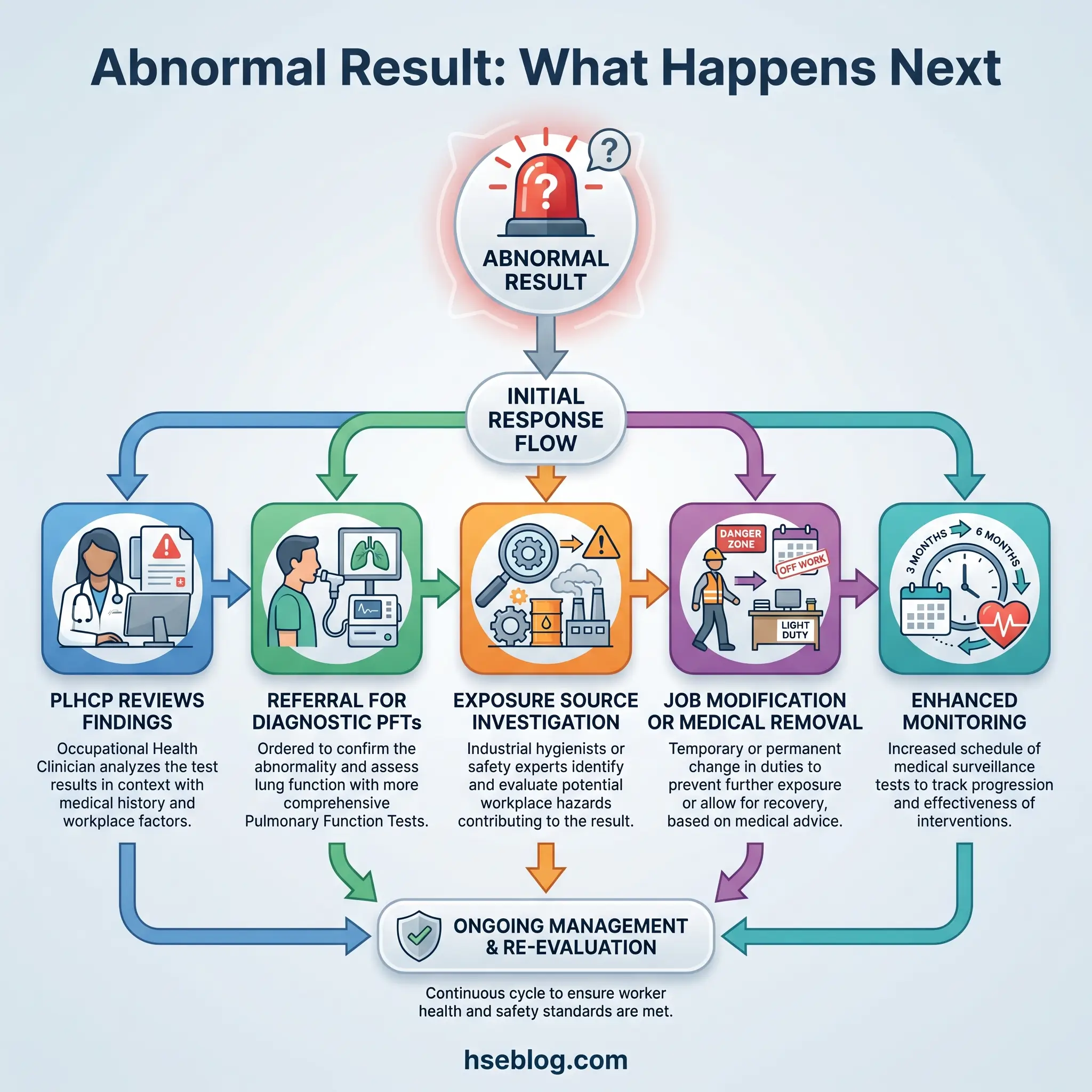
Individual test quality is necessary but not sufficient. A programme that produces Grade A spirometry on each session but has no system for reviewing trends across the workforce, auditing technician performance, or validating equipment over time is a programme built on sand.
The qualified reviewer — typically a physician with spirometry interpretation expertise — should perform QA over-reads of spirometry tracings. This is not the same as the PLHCP reviewing results for medical decisions. The over-reader is checking data quality: Are the curves smooth and physiologically plausible? Is the peak flow sharp or hesitant? Does the volume-time curve show a true plateau or was the exhalation terminated early? Were the quality grades assigned correctly?
Common sources of error that QA review catches include zero-flow errors (where the spirometer’s baseline drifts, adding volume before the worker begins exhaling — this creates a falsely obstructive pattern), volume drift from temperature changes, slow starts that blunt the peak flow, submaximal effort producing reproducible but effort-dependent curves, and cough artifacts within the first second that reduce the measured FEV1.
A written procedure manual is not optional. It documents the testing protocol, equipment maintenance schedule, calibration verification procedures, interpretation criteria, referral pathways, and the qualifications of every person involved. When an auditor asks to see your spirometry procedure, “we follow OSHA 3637” is not an answer. The manual must be site-specific.
The Fix That Works: Review aggregate spirometry data across your workforce quarterly. If one job category shows a higher rate of declined FEV1 than others, that is not a coincidence — it is an exposure signal that should trigger an industrial hygiene review before the next round of spirometry.
Recordkeeping and Data Retention Obligations
A spirometry result without a record is a spirometry result that never happened — at least from a compliance and legal perspective. The obligations are specific and non-negotiable.
Under OSHA’s Access to Employee Exposure and Medical Records standard (29 CFR 1910.1020), medical records including spirometry results must be retained for the duration of employment plus 30 years. This means that a worker who spent five years at a facility and left in 2025 must have their spirometry records available until 2060.
COSHH health surveillance records carry an even longer obligation: at least 40 years from the date of the last entry. Given that mesothelioma latency can exceed 40 years and accelerated silicosis cases have emerged decades after exposure, these retention periods are not arbitrary bureaucracy — they are evidence preservation.
| Jurisdiction / Standard | Retention Period | What Must Be Recorded |
|---|---|---|
| OSHA 29 CFR 1910.1020 | Employment + 30 years | Test date, worker ID, spirometer used, calibration log, FVC, FEV1, FEV1/FVC, curves, technician ID, quality grade |
| COSHH (UK) | At least 40 years | Surveillance dates, results, health professional identity, exposure context |
| MSHA 2024 Silica Rule | Employment + 6 months | Medical surveillance records including spirometry |
Confidentiality is a regulatory requirement, not a courtesy. The worker’s spirometry results and clinical data go to the PLHCP. The employer receives a written medical opinion stating whether the worker is fit for their assigned duties and any recommended limitations — but not the FEV1 value, not the curves, not the diagnosis. Workers have the right to access their own records at any time and at no cost.
Industries and Exposures That Trigger Spirometry Requirements
Knowing which regulation applies is step one. Knowing whether your workplace falls under that regulation requires mapping your exposures to the regulatory framework. The re-emergence of accelerated silicosis cases linked to engineered stone countertop fabrication — a global trend documented since 2019 and still accelerating — demonstrates that spirometry triggers can emerge in industries that did not previously consider themselves high-risk.
- Mineral dusts: Respirable crystalline silica exposure in construction, mining, tunnelling, stone countertop fabrication, foundry work, and glass manufacturing triggers spirometry under OSHA’s silica standards. Asbestos exposure during demolition, renovation, maintenance, and insulation work triggers testing under the asbestos standard. Coal dust exposure in mining falls under the new MSHA 2024 rule.
- Metal fumes: Welding generates fumes containing manganese, chromium, nickel, and other metals. Cadmium processing (battery manufacturing, smelting, plating) triggers spirometry under the cadmium standard. Beryllium machining triggers surveillance under the beryllium standard.
- Organic dusts: Cotton dust in textile manufacturing falls under OSHA’s most technically detailed spirometry standard. Wood dust in furniture making, carpentry, and sawmilling creates exposure to known respiratory sensitisers. Grain dust in agricultural processing and bakery flour dust trigger COSHH health surveillance requirements.
- Chemical vapours: Formaldehyde in laboratories and manufacturing, benzene in petrochemical operations, and isocyanates in spray painting, polyurethane production, and coatings application all trigger spirometry obligations.
- Respiratory sensitisers (COSHH): Latex in healthcare, enzyme-containing products in pharmaceutical and cleaning product manufacturing, and reactive dyes in textile dyeing operations all require spirometry-inclusive health surveillance when exposure cannot be fully controlled.
Based on UK Labour Force Survey data averaged across 2022/23–2024/25, approximately 22,000 new cases occur each year of breathing or lung problems that workers believe were caused or worsened by work. Chronic respiratory diseases — excluding lung cancer and infections — are the third leading cause of death globally, responsible for 4 million deaths and 103.5 million disability-adjusted life years worldwide in 2019. These figures frame spirometry not as an administrative burden but as a frontline detection system for a global health crisis rooted substantially in workplaces.

Infection Control and Post-Pandemic Spirometry Considerations
Spirometry is classified as an aerosol-generating procedure. A forced maximal exhalation propels respiratory droplets and aerosol particles across the testing space. This reality, always present but underappreciated before 2020, has permanently changed how occupational spirometry sessions should be managed.
Disposable single-use mouthpieces and bacterial/viral filters are baseline requirements. Between each worker, the mouthpiece-filter assembly is discarded and external spirometer surfaces are disinfected. The testing room should have adequate ventilation — ideally mechanical ventilation providing at least six air changes per hour, with time allowed between workers for aerosol clearance. Spatial distancing between the technician and the worker during the forced exhalation maneuver reduces operator exposure.
Emerging technologies are addressing this challenge. Bluetooth-enabled spirometers allow the technician to coach and monitor test quality from a distance, maintaining the critical real-time curve assessment without standing at arm’s length from the forced exhalation. Non-contact respiratory monitoring methods are under development but have not yet reached the accuracy required for occupational screening against regulatory standards.
Return-to-work spirometry for workers recovering from significant respiratory illness — including post-viral syndromes — is becoming a standard component of fitness-for-duty assessments. A worker returning to a silica-exposed role after a prolonged respiratory illness needs updated baseline spirometry. The pre-illness baseline may no longer be representative, and subsequent longitudinal monitoring should account for any infection-related decline.
Common Mistakes and How to Avoid Them
Technical errors and programme management failures erode spirometry data quality in predictable patterns. Identifying them is straightforward. Eliminating them requires systematic attention.
- Using spirometry as a substitute for exposure control. Spirometry detects damage — it does not prevent it. I have heard the argument: “We test every year, so we’re covered.” Testing frequency does not reduce airborne silica concentration. Spirometry sits downstream of the hierarchy of controls, not in place of it. Engineering controls, ventilation, substitution, and administrative measures reduce exposure. Spirometry confirms whether those measures are working.
- Failing to maintain calibration verification logs. The calibration syringe sits on the shelf. The log book has entries from last Tuesday and a gap before that stretching back three weeks. In between, forty workers were tested with an unverified instrument. Every result from those sessions is indefensible.
- Inadequate worker coaching. A technician who does not demonstrate the maneuver, does not encourage during exhalation, and does not explain why the test matters will produce submaximal effort. “Blow into this” is not coaching. “Take the biggest breath of your life, seal your lips around the mouthpiece, and blast it out like you’re blowing out a hundred candles — and keep blowing until I tell you to stop” — that is coaching.
- Relying solely on cross-sectional interpretation. A programme that compares each session only to population predicted values will miss the worker whose FEV1 dropped from 4.5 liters to 3.6 liters over six years — still above the LLN, still flagged as “normal,” still losing lung function at three times the expected rate.
- Not accounting for pre-test factors. The worker who jogged to the testing trailer, or who smoked fifteen minutes before the session, or whose coveralls are cinched tight across the chest, is not producing physiologically valid data. Every pre-test checklist item exists because it affects the measurement.
- Overlooking aggregate workforce analysis. Individual results are trees. The forest is the pattern across job categories, exposure zones, and tenure cohorts. A spike in borderline-abnormal results among workers in one department is not a spirometry problem — it is an exposure problem waiting for someone to notice.
Frequently Asked Questions
The industry’s most persistent error with workplace spirometry is treating it as a compliance checkbox rather than a clinical surveillance system. The companies that get it right do not simply schedule annual tests and file the results. They maintain calibrated equipment with documented verification. They employ or contract NIOSH-certified technicians who coach, not just record. They track every worker’s FEV1 longitudinally against a personal baseline, watching for the slope of decline that cross-sectional testing alone would miss. And they use aggregate data to ask the harder question: is our exposure control programme actually working, or is spirometry just documenting the damage?
The single highest-impact change most occupational spirometry programmes can make is implementing systematic longitudinal tracking. Cross-sectional interpretation is necessary, but it is the longitudinal trend — plotted over three, five, ten years — that catches the slow theft of lung function before it becomes irreversible disease. A worker’s FEV1 today means little in isolation. That same FEV1 plotted against a baseline from three years ago tells a story that can change the ending.
Every forced exhalation into that mouthpiece is a data point. The question is whether your programme has the systems to make that data point matter.

