TL;DR
- CO becomes dangerous below levels you can smell or see: Concentrations as low as 50 ppm sustained over 8 hours cause measurable health damage, yet carbon monoxide is completely odorless and colorless.
- OSHA’s permissible exposure limit is 50 ppm TWA: Exceeding this in any work environment requires immediate ventilation, atmospheric monitoring, and potential evacuation.
- Concentrations above 400 ppm are immediately life-threatening: At this level, headaches and confusion escalate within minutes, and collapse can follow within two hours.
- Continuous atmospheric monitoring is non-negotiable: Single-point readings miss CO buildup patterns — real-time personal monitors catch lethal trends before symptoms start.
- Every CO exposure incident traces back to predictable failures: Poor ventilation, unserviced combustion equipment, and absent detection systems account for the vast majority of fatalities.
The four-gas detector on the entry attendant’s belt started alarming thirty seconds after the maintenance crew entered the boiler room. Carbon monoxide: 87 ppm and climbing. The crew had been inside for less than a minute. No one felt anything yet — no headache, no dizziness. The gas had been building silently for hours from a faulty flue damper. I ordered an immediate evacuation and locked out the space. Post-incident testing revealed the CO concentration had peaked at 310 ppm near the ceiling. Had the detector not been calibrated the previous morning, three workers would have continued their task in an invisible atmosphere that was quietly poisoning them.
Carbon monoxide levels define the line between a safe work environment and a body recovery operation. CO kills because it is perfectly designed to be ignored — no color, no odor, no irritation. Understanding the specific concentrations that separate safe exposure from organ damage, incapacitation, and death is not academic. It is the foundation of every atmospheric monitoring program, every confined space entry permit, and every combustion equipment maintenance schedule. This article breaks down exactly what carbon monoxide levels mean in parts per million, how to interpret them in real operations, and what controls prevent exposure from crossing the line.

What Carbon Monoxide Is and Why It Kills Silently
Carbon monoxide is a gas produced by incomplete combustion of any carbon-based fuel — natural gas, propane, diesel, gasoline, wood, coal, or kerosene. It shares a critical physical trait with breathable air: it is roughly the same density, meaning it distributes evenly throughout any enclosed or semi-enclosed space rather than settling predictably at floor or ceiling level.
The lethality mechanism is straightforward but ruthless. CO binds to hemoglobin in blood approximately 200–250 times more aggressively than oxygen does. When you inhale carbon monoxide, it locks onto the same receptor sites that normally carry oxygen to your brain, heart, and muscles. The result is a condition called carboxyhemoglobin (COHb) saturation — your blood is physically full, but it is carrying poison instead of oxygen. Your body suffocates from the inside while you continue breathing normally.
This binding affinity is what makes carbon monoxide levels so critical to understand. The damage is cumulative, dose-dependent, and time-dependent. A concentration that feels harmless for ten minutes can cause brain injury over two hours. The relationship between parts per million in the air and carboxyhemoglobin percentage in blood follows a predictable curve — and once COHb saturation exceeds 40%, cardiac arrest and death become probable.
I have investigated CO exposure incidents where workers described feeling “a little tired” at concentrations that were already above 100 ppm. By the time confusion sets in, the victim often lacks the cognitive ability to recognize the danger or walk themselves out. This is why we never rely on symptoms as a detection method. Instruments detect carbon monoxide. People detect consequences.
Occupational Exposure Limits for Carbon Monoxide
Regulatory bodies worldwide set carbon monoxide levels based on time-weighted averages and ceiling limits. These thresholds represent the maximum allowable concentration before engineering controls, respiratory protection, or evacuation become mandatory. The differences between standards matter — and applying the wrong one can leave workers exposed above safe levels while technically “compliant” with a weaker standard.
The following table compares the primary occupational exposure limits for carbon monoxide across major regulatory frameworks:
| Regulatory Body | TWA (8-hour) | STEL (15-min) | Ceiling / IDLH |
|---|---|---|---|
| OSHA PEL | 50 ppm | Not established | — |
| NIOSH REL | 35 ppm | 200 ppm (ceiling) | 1,200 ppm (IDLH) |
| ACGIH TLV | 25 ppm | Not established | — |
| HSE UK WEL | 20 ppm | 100 ppm | — |
OSHA’s permissible exposure limit of 50 ppm as an 8-hour TWA has not been updated in decades and is widely recognized within the industrial hygiene community as insufficiently protective. NIOSH recommends a lower limit of 35 ppm, and the ACGIH threshold limit value of 25 ppm reflects more current toxicological evidence. When I develop exposure monitoring programs, I default to the ACGIH TLV of 25 ppm as the operational standard — not the OSHA PEL.
Pro Tip: When clients or site managers push back on using stricter limits than OSHA requires, I point to the ACGIH TLV documentation and the growing body of evidence on chronic low-level CO effects, including cognitive impairment and cardiovascular stress at concentrations well below 50 ppm. Regulatory compliance is a floor, not a ceiling.
The HSE UK workplace exposure limit of 20 ppm long-term and 100 ppm short-term represents the most conservative enforceable standard among major jurisdictions. For multinational operations — especially EPC projects that cross regulatory boundaries — I recommend adopting the HSE UK WEL as the baseline, which exceeds OSHA’s threshold and provides the greatest margin of safety.

Carbon Monoxide Concentration Ranges: What Each Level Means
Understanding what happens to the human body at specific carbon monoxide levels is the difference between reading a number on a monitor and knowing whether to ventilate, evacuate, or call for medical assistance. The following breakdown covers the concentration ranges that matter most in field operations.
0–9 ppm: Normal Ambient Background
Outdoor ambient air typically contains 0.1–0.5 ppm of carbon monoxide from natural sources and distant combustion. Indoor environments with properly functioning gas appliances may register 5–9 ppm without indicating a hazard. Readings in this range during atmospheric monitoring confirm normal conditions.
However, a “normal” reading should never generate complacency. I have seen teams accept baseline readings of 8–9 ppm without questioning why the background was that high in a space with no obvious combustion source. In one case, the elevated background turned out to be exhaust infiltration from a diesel generator positioned upwind of an air intake — a systemic issue that would have gone unnoticed without trending data over multiple shifts.
10–25 ppm: Caution — Investigate the Source
At 10–25 ppm, carbon monoxide is present above typical background and warrants immediate source identification. For healthy adults, short-term exposure at these levels is unlikely to cause acute symptoms, but prolonged exposure (8+ hours daily over weeks) falls within the range where the ACGIH TLV triggers mandatory controls.
Key actions at this concentration range are critical to preventing further escalation:
- Identify the combustion source: Check all fuel-burning equipment, engines, heaters, and generators within or adjacent to the monitored area
- Verify ventilation rates: Confirm that mechanical or natural ventilation is operating as designed — fan speeds, damper positions, duct obstructions
- Review CO trending data: Compare current readings to historical baselines for the same area and shift conditions
- Notify the area supervisor: Document the elevated reading and corrective actions in the shift log
26–50 ppm: Elevated — Controls Required
This range sits between the ACGIH TLV and the OSHA PEL. Workers with cardiovascular conditions, pregnant workers, or those performing heavy physical labor may experience measurable health effects at the lower end of this band. Carboxyhemoglobin levels begin rising above the 3–5% range associated with impaired exercise tolerance and early cardiovascular stress.
Operational responses at this level should include active ventilation improvement and investigation:
- Increase ventilation immediately: Open additional air pathways, reposition portable fans, or adjust HVAC supply to dilute CO concentration
- Switch to continuous monitoring: Move from spot checks to real-time alarming monitors clipped to the breathing zone
- Restrict exposure duration: Reduce work periods in the affected area and rotate personnel to limit individual dose accumulation
- Evaluate the need for respiratory protection: If ventilation cannot reduce levels below 25 ppm within 30 minutes, consider supplied-air or air-purifying respirators rated for CO
Pro Tip: Standard air-purifying respirators with organic vapor cartridges do not protect against carbon monoxide. CO requires a specific CO-rated catalytic cartridge or, more reliably, supplied-air breathing apparatus. I have pulled workers out of hazardous atmospheres wearing the wrong cartridge type — convinced they were protected when their respirator was doing nothing for them.
51–200 ppm: Dangerous — Evacuate and Ventilate
Carbon monoxide levels between 51 and 200 ppm produce symptoms in most adults within one to three hours. The progression is well-documented and dose-dependent:
- 50–100 ppm: Mild headache after 2–3 hours of continuous exposure; reduced concentration and reaction time
- 100–150 ppm: Throbbing headache within 1–2 hours; dizziness, nausea, and impaired judgment
- 150–200 ppm: Headache within 1 hour; confusion, disorientation, and muscle weakness; risk of collapse during physical exertion
At any reading above 50 ppm, I stop the job. Full stop. There is no task urgent enough to continue breathing an atmosphere at this concentration without supplied-air respiratory protection and a defined rescue plan. Workers exposed at this range need fresh air, medical evaluation, and COHb blood testing before being cleared to return.
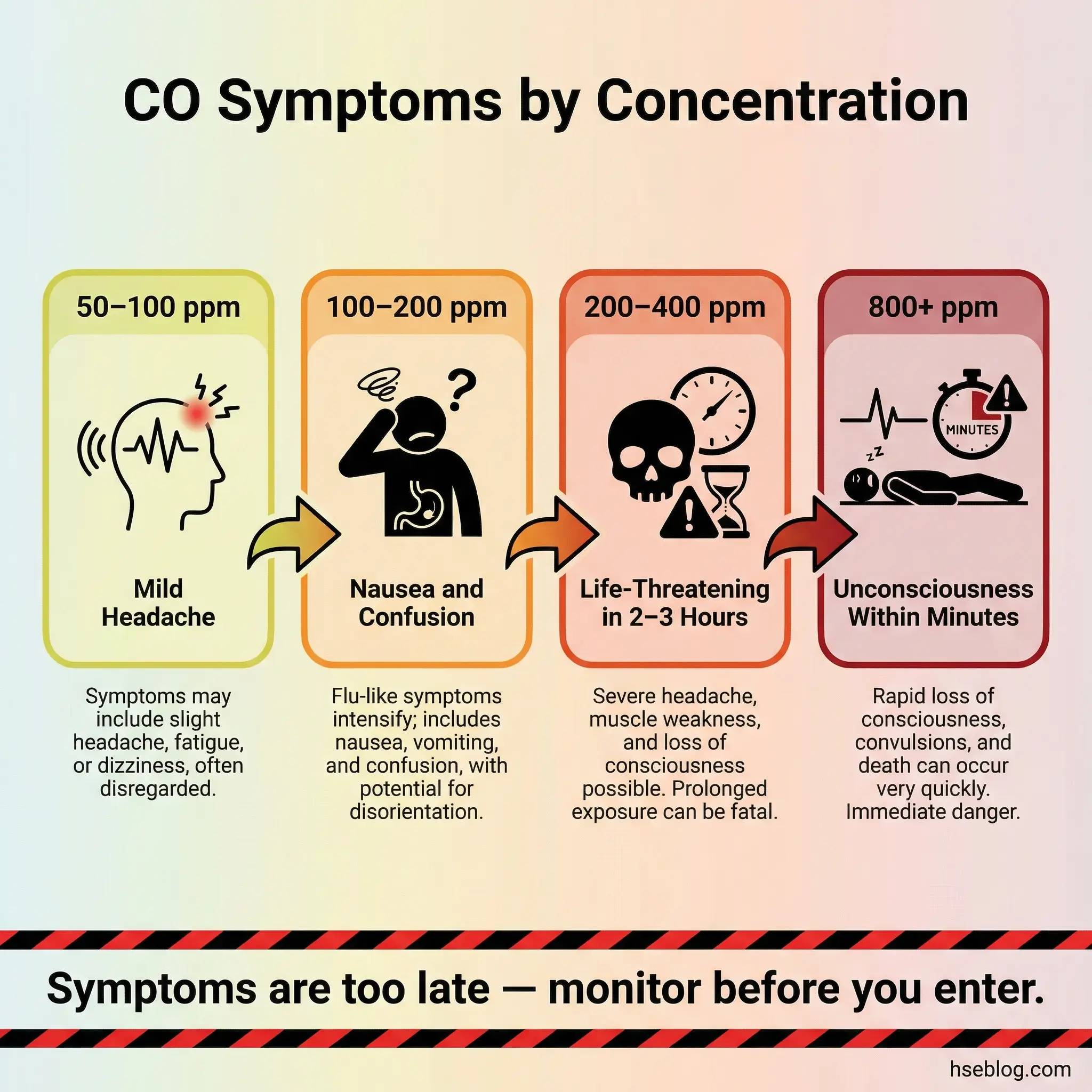
200–400 ppm: Life-Threatening — Immediate Evacuation
Concentrations in this range cause life-threatening symptoms within two to three hours. Headache becomes severe within 30 minutes. Judgment and motor coordination deteriorate rapidly. Workers performing physical tasks — climbing, lifting, operating equipment — face elevated risk of falls, dropped loads, and uncontrolled movements because their muscles are being starved of oxygen even while they feel like they are breathing normally.
During one confined space rescue drill that turned into an actual response, we recovered a welder from a partially enclosed pipe rack where CO had accumulated to 340 ppm. The welder had been working for approximately 45 minutes before his buddy noticed slurred speech and slow movements. His COHb level at the hospital measured 28%. He survived, but neurological follow-up three months later revealed measurable short-term memory deficits.
400 ppm and Above: Immediately Dangerous to Life
NIOSH classifies 1,200 ppm as the Immediately Dangerous to Life or Health (IDLH) concentration for carbon monoxide. But functional incapacitation begins well before that threshold. At 400 ppm, most adults experience life-threatening symptoms within two hours. At 800 ppm, unconsciousness can occur within minutes. Above 1,600 ppm, death follows rapidly — sometimes within 20 minutes.
The critical operational reality at these concentrations is sobering:
- No entry without SCBA: Self-contained breathing apparatus is the only acceptable respiratory protection at or above IDLH levels
- No solo rescues: Would-be rescuers who enter without SCBA become additional victims — this is the single most common cause of multiple-fatality CO incidents
- Atmospheric isolation: Identify and shut down the CO source before any rescue attempt unless life is in immediate danger
- Emergency medical response: All exposed workers require immediate transport, 100% oxygen therapy, and COHb blood testing
NIOSH defines the IDLH for carbon monoxide at 1,200 ppm — but impairment, collapse, and death occur at concentrations far below this threshold. Never treat the IDLH as the point where danger begins. Danger begins the moment your monitor reads above background.
Common Sources of Carbon Monoxide in the Workplace
Understanding where carbon monoxide comes from in real operations is the foundation of source control — the most effective layer of the hierarchy of control. Every CO exposure I have investigated traced back to one of a limited number of source categories, and most were predictable and preventable.
The following sources represent the most common CO generators across industrial and construction environments:
- Internal combustion engines in enclosed or semi-enclosed areas: Forklifts, generators, concrete saws, compactors, and vehicles operated in warehouses, tunnels, underground car parks, and partially enclosed loading docks — this is the single largest source of occupational CO exposure
- Propane and natural gas-fired equipment: Heaters, furnaces, ovens, and dryers with cracked heat exchangers, blocked flues, or inadequate combustion air supply
- Welding and cutting operations: Oxy-fuel cutting, gas welding, and certain shielded metal arc welding processes produce CO as a byproduct, particularly in confined or poorly ventilated spaces
- Diesel exhaust in enclosed structures: Construction sites using diesel-powered pumps, compressors, or generators inside buildings under construction — before permanent ventilation is operational
- Industrial process emissions: Blast furnaces, coke ovens, catalytic crackers, and syngas operations generate CO as a process byproduct at concentrations that can be immediately lethal
- Fire or smoldering combustion: Post-fire environments, hot work near combustible materials, and overheated electrical equipment can produce CO without visible flame
Pro Tip: The most dangerous CO exposures I have seen were not from obvious sources like running engines. They came from hidden ones — a cracked heat exchanger in a roof-mounted unit pushing CO into an office space through the HVAC system, exhaust from a neighboring tenant’s generator infiltrating through shared wall penetrations, or a blocked flue on a rarely inspected water heater. Always investigate the non-obvious sources first.

How to Monitor Carbon Monoxide Levels Effectively
Atmospheric monitoring for carbon monoxide is only as reliable as the equipment, calibration, placement, and interpretation behind it. I have reviewed monitoring programs that ticked every compliance box on paper but failed catastrophically in practice because the fundamentals were wrong.
Effective CO monitoring requires discipline in both equipment selection and operational practice:
- Personal single-gas or multi-gas monitors: Clip-on electrochemical sensors worn in the breathing zone provide real-time exposure data for individual workers — these are the frontline defense for any task involving combustion sources or enclosed areas
- Fixed-point area monitors: Wall-mounted or ceiling-mounted sensors in boiler rooms, enclosed parking structures, generator rooms, and process areas provide continuous area surveillance and feed into building management systems
- Pre-entry atmospheric testing: Before any confined space entry, test the atmosphere at multiple levels (top, middle, bottom) with a calibrated instrument — a single point reading is insufficient because CO distributes based on air currents, not just density
- Continuous monitoring during occupancy: Pre-entry testing alone is not enough — conditions change as work progresses, particularly when hot work, engine operation, or chemical processes are active inside the space
Calibration and maintenance failures undermine even the best monitoring hardware. These common mistakes turn expensive instruments into false assurance:
- Bump testing skipped before each shift: A bump test confirms the sensor responds to a known concentration of target gas — without it, you cannot confirm the instrument is functional, only that it powers on
- Expired or cross-contaminated calibration gas: Using gas cylinders past their expiration date or cylinders that have been stored improperly produces unreliable calibration baselines
- Sensor drift not tracked: Electrochemical CO sensors degrade over time and can under-read by 10–30% near end of life — track sensor age and replace on schedule, not on failure
- Instrument stored in contaminated environments: Detectors left overnight in areas with residual CO, solvent vapors, or hydrogen sulfide can have saturated sensors at the start of the next shift, causing delayed or suppressed alarms
OSHA 29 CFR 1910.146 requires continuous atmospheric monitoring during confined space entry when hazardous atmospheres may develop. A single pre-entry reading does not satisfy this requirement when conditions can change during the work.
Carbon Monoxide Exposure: Site-Level Mistakes That Kill
Every fatal CO exposure I have investigated shared common organizational and behavioral failures. These are not obscure edge cases. They are the same mistakes repeated across industries, geographies, and decades — and they remain the primary cause of preventable deaths.
The following mistakes appear with disturbing consistency in CO incident investigation reports:
- Running combustion equipment indoors “just for a few minutes”: Portable generators, propane heaters, and gasoline-powered tools operated inside garages, basements, and partially enclosed structures — CO reaches dangerous levels within minutes in small volumes, not hours
- Assuming natural ventilation is sufficient: Open doors and windows do not guarantee adequate air exchange — wind direction, temperature differentials, and physical obstructions all affect actual ventilation rates
- Relying on smell or symptoms to detect CO: Carbon monoxide is odorless and colorless — by the time a worker feels symptoms, they may already have a COHb level above 15%, with impaired judgment making self-rescue unreliable
- Attempting unprotected rescue of CO victims: The impulse to rush in and pull out a collapsed colleague without respiratory protection is understandable — and it is the single most common cause of multiple-fatality confined space incidents
- Ignoring low-level alarms: Treating a detector alarm at 25 or 35 ppm as a nuisance rather than an investigation trigger allows conditions to escalate while attention drifts elsewhere
- Failing to test adjacent spaces: CO migrates through wall penetrations, shared ductwork, floor drains, and pipe chases — a safe reading in one room does not guarantee the room next to it is clear
Pro Tip: During incident investigations, I always ask: “What was the alarm response procedure, and when was it last drilled?” In most fatal cases, the procedure either did not exist, existed only on paper, or had never been practiced with the actual people working in the actual space. A procedure that has not been drilled is not a procedure — it is a document.

Practical Control Measures for Carbon Monoxide Exposure
Controlling carbon monoxide exposure follows the hierarchy of control — and the most effective measures address the source before they address the worker. Engineering controls that eliminate or reduce CO generation outperform PPE every time, because PPE fails the moment it is removed, improperly fitted, or selected incorrectly.
Elimination and Substitution
The most reliable way to prevent CO exposure is to remove the combustion source entirely or replace it with an alternative that does not produce carbon monoxide:
- Electric-powered equipment over combustion-driven alternatives: Battery-electric forklifts in warehouses, electric compressors in enclosed spaces, and corded power tools instead of gasoline-powered versions eliminate CO generation at the source
- Relocate combustion equipment outdoors: Position generators, compressors, and heaters outside enclosed work areas with exhaust directed away from air intakes, doorways, and windows
- Replace propane heaters with electric radiant heating: In construction enclosures and temporary shelters, electric radiant panels eliminate combustion byproducts entirely
Engineering Controls
When combustion sources cannot be eliminated, engineering controls reduce CO concentration to below occupational exposure limits:
- Mechanical ventilation systems: Calculate required air exchange rates based on the CO generation rate of the equipment in use — do not guess ventilation requirements based on room size alone
- Local exhaust ventilation on engine exhaust: Direct-connect tailpipe extraction systems on forklifts, vehicles, and generators capture CO at the source before it disperses into the breathing zone
- CO-linked ventilation interlocks: Fixed monitoring systems connected to ventilation fan controls that automatically increase air exchange when CO levels rise above a setpoint — standard practice in enclosed parking structures and vehicle maintenance bays
- Combustion equipment maintenance programs: Scheduled inspection and servicing of heat exchangers, burner assemblies, flue systems, and catalytic converters prevents the degradation that causes incomplete combustion and elevated CO output
Administrative Controls and PPE
Administrative controls and respiratory protective equipment serve as supplementary layers when engineering controls alone cannot maintain CO below exposure limits:
- Atmospheric monitoring programs: Continuous personal and area monitoring with documented alarm setpoints, response procedures, and escalation criteria
- Work duration limits: Restrict time in areas with measurable CO to maintain TWA below the applicable OEL — use exposure calculations based on actual concentration and duration, not assumptions
- Training on CO recognition and response: Every worker in a CO-risk area must understand what the monitor alarm means, where to evacuate, and why they must never attempt an unprotected rescue
- Respiratory protection selection: For short-duration tasks in CO atmospheres below IDLH, CO-specific catalytic cartridges on air-purifying respirators provide protection — but supplied-air systems or SCBA are mandatory at or above IDLH concentrations
Carbon Monoxide in Confined Spaces: A Compounded Risk
Confined spaces magnify every aspect of carbon monoxide danger. Limited volume means small CO sources produce high concentrations quickly. Restricted openings limit natural air exchange. Difficult access complicates rescue. I have responded to more confined space CO incidents than any other single atmospheric hazard, and the pattern is always the same: inadequate pre-entry testing, absent continuous monitoring, and no rescue plan that accounts for an irrespirable atmosphere.
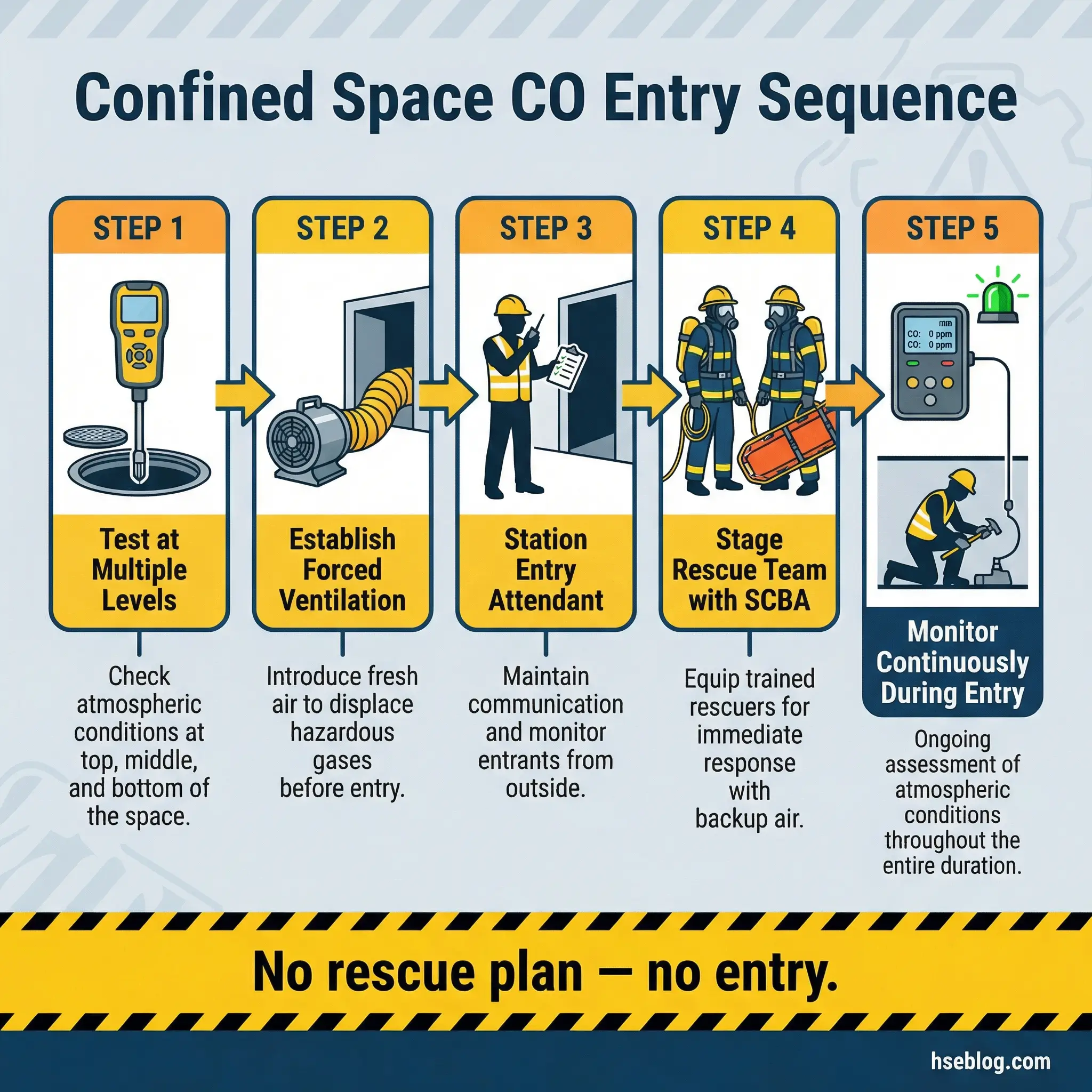
The confined space entry procedure must account for carbon monoxide as a primary atmospheric hazard in any space where combustion has occurred, fuel-burning equipment operates nearby, or organic decomposition produces CO:
- Test the atmosphere at multiple levels before entry using a calibrated four-gas detector that includes a CO sensor — record readings at the top, middle, and bottom of the space
- Establish mechanical ventilation capable of maintaining CO below 25 ppm (ACGIH TLV) throughout the entry duration — verify with continuous monitoring
- Position the entry attendant with a direct line of communication to entrants and to emergency services — the attendant must have the authority to order immediate evacuation
- Deploy a rescue team with SCBA staged at the entry point before any worker enters — never plan to “figure out rescue” after an incident occurs
- Monitor continuously throughout the entry and log readings at defined intervals — any upward trend in CO requires investigation, not just a check against the alarm threshold
The relationship between carbon monoxide and confined spaces accounts for a disproportionate share of occupational CO fatalities. In confined space incidents, would-be rescuers who enter without respiratory protection account for the majority of additional deaths — a fact that reinforces why rescue planning is not optional.
Conclusion
Carbon monoxide levels are not abstract numbers on a regulatory table. They are the dividing line between a normal shift and a fatality investigation. The difference between 25 ppm and 200 ppm is the difference between a healthy worker going home and a family receiving a phone call no one should have to make. Every control measure discussed in this article — source elimination, engineering ventilation, continuous atmospheric monitoring, proper respiratory protection, and trained emergency response — exists because someone, somewhere, died from its absence.
Prevention works when it is built into systems, not left to individual awareness. A worker cannot smell carbon monoxide, cannot see it, and by the time they feel its effects, their ability to save themselves may already be compromised. The instrumentation, the calibration discipline, the ventilation calculations, the rescue staging — these are the defenses that stand between routine operations and catastrophe.
The standard I hold on every site I manage is simple: if you cannot prove the atmosphere is safe with a calibrated instrument and a documented reading, you do not enter. No schedule pressure, no production target, and no client demand justifies sending a person into a space where carbon monoxide levels are unknown. The numbers on the detector are not suggestions — they are the last warning you will get.

